Link on the site here
Introduced in 2018 for cyproterone acetate (Androcur®) and then in 2020 for chlormadinone acetate (Lutéran®) and for nomestrol acetate (Lutényl®), brain MRI meningioma screening in patients exposed to progestins is one of the imagery requests with which the radiologists are confronted in everyday life. We will return here to the literature at the origin of this screening, its practical modalities, the type of discovery tumors and their management.
Progestative treatments have anti -diand and progestogen activity with framed indications (severe hirsutism, endometriosis, polycystic ovary syndrome, etc.). Two French epidemiological studies 1.2 were carried out from the national health data system by identifying progestogen users and patients with intracranial meningioma. These studies reported an increased risk of linked meningiomas: 1/ to the progestin received ( more important in androcur® users than Lotényl®/ Lotéran® ), 2/ at the dose and the duration of exposure to treatment and 3/ at patient age. These observations are at the origin of the systematic meningioma screening implemented by the National Health and Medicine Agency.
Apart from symptomatic patients in whom brain imaging should always be carried out regardless of hormonal exposure, screening for asymptomatic patients is organized as follows:
- For Androcur®, a brain MRI is systematically required before the initiation of treatment (the presence of a meningioma contributing the latter). Then, if the treatment is continued, surveillance will include a brain MRI at 5 years then every 2 years if the MRI at 5 years is normal. The “microdosted” treatments in cyproterone acetate - like Diane35® - do not have a meningioma ride and are therefore excluded from screening.
- For Lutényl®/Lutéran®, a brain MRI will only be carried out before treatment starts in the event of specific risk factors (brain radiotherapy in childhood or type 2 neurofibromatosis). Subsequently, if the treatment is continued, regardless of the patient's risk factors, surveillance will include an MRI at 1 year, then at 5 years then every 2 years if the MRI at 5 years is normal.
In patients who have stopped progestin treatment - whatever it is - and not having symptoms, it is not recommended to make brain imaging.
Regarding the methods of screening, privileged imaging is brain MRI with gadolinium injection given the absence of ionizing radiation and the finer analysis of the extra-axial structures it allows. Building on the experience of our center, we offer a rapid exploration protocol consisting of an injection of a gadolini contrast agent in the cabin, then the realization of a T2 Flair and 3D T1 3 . Given the small size of the meningiomas discovered in the context of systematic screening, we advise to favor sequences echoing spin with saturation of the fat signal rather than the gradient echo. Thus, you will educate your exam in particular near the vein structures. Other sequences will complete the protocol, according to symptoms reported by patients.
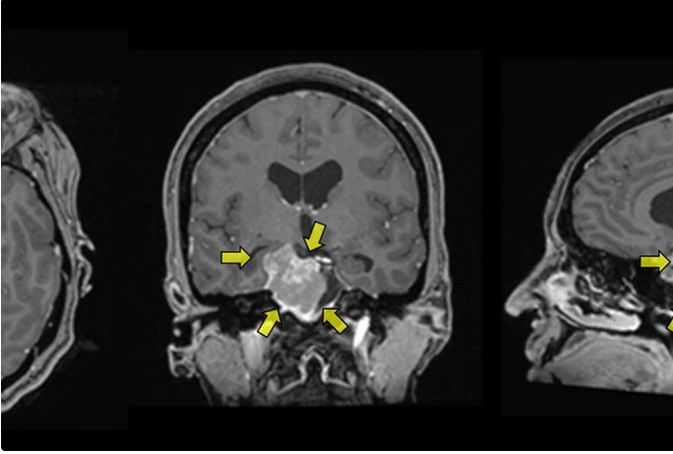
The meningiomas associated with progestins differ little from meningiomas discovered in the general population. These are extra-axial tumors based on Dure-Merian implantation strongly enhanced by the contrast, sometimes associated with hyperostosis opposite. However, asymptomatic tumors detected in the event of exposure to progestins are often multiple and small size 3 . Particular attention should be paid to the base of the skull where small tumors can quickly become symptomatic, especially near the nervous foramens. When a meningioma is discovered in a patient exposed to the progestins, the patient should be sent to neurosurgery for a specialized opinion. These tumors being for the most part hormone-dependent, the suspension of the progestogen induces in the majority of cases a spontaneous volume stability or decay (see image), which makes it possible to overcome an invasive 3.4 . The pace of monitoring of this decrease is not consensual and will be adapted on a case -by -case basis.
References
1. Weill, A. et al. Use of High dose Cyproterone Acetate and Risk of Intracranial Meningioma in Women: Cohort Study. BMJ 37, 1–13 (2021).
2. Hoisnard, L. et al. Risk of Intracranial Meningioma With Three Potent Progestogens: A Population - Based Case - Control Study. Euro J of Neurology ENE.15423 (2022) DOI: 10.1111/ENE.15423.
3. Samoyeau, T. et al. Meningioma in Patients Exposed to Progestin Drugs: Results from A Real-Life Screening Program. J Neurooncol 160, 127–136 (2022).
4. Malaize, H. et al. Evolution of the Neurosurgical Management of Progestin-Associated Meningiomas: A 23-Year Single-Center Experience. J Neurooncol 152, 279–288 (2021).
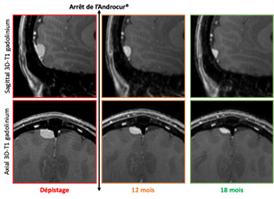
Spontaneous decrease of meningioma after interruption of exposure to cyproterone acetate in a 28 -year -old, asymptomatic patient, exposed for 5 years to Androcur®. T1 3D sequences with gadolinium injection.
Dr. Thomas Samoyeau , Paris Cité
Pr. Johan Pallud , Service of Neurosurgery, GHU Paris Psychiatry and Neuroscience
Dr. Corentin Provost , Diagnostic and Therapeutic Imagery Service, GHU Paris Psychiatry & Neuroscience
Pr. Catherine Oppenheim , Diagnostic and therapeutic imaging service, GHU Neuroscience
Dr. Joseph Benzakoun , Diagnostic and therapeutic imaging service, GHU Paris Psychiatry & Neuroscience