The progestogen pills Lutéran, Lotényl and their generics would increase the risk of developing a brain tumor. In order to establish new recommendations for healthcare professionals and patients, ANSM has scheduled a public consultation on YouTube on November 2. In the meantime, women wonder: have they endangered?
"I believed in a fake news and when I saw that it was growing, I started to worry." Léa*, under Lutéran for three years, like thousands of other women, learned only a few weeks ago on social networks that his pill could lead to an over-risk to develop a meningioma, a brain tumor , benign in most cases. Today, the National Medicines Safety Agency (ANSM) estimates that 600,000 the number of women exhibited every year at Lutéran, Lotényl and their generics . "This treatment can be taken at different times in its hormonal life," said the agency in Neon. These two progestin drugs, that is to say without estrogen, are notably indicated in cases of gynecological disorders such as endometriosis , fibroid, painful rules or even pre-menopause.
The 24 -year -old student had the right reflex: calling her gynecologist without delay. "I came across the secretary, she looked a little overwhelmed," continues the young woman from Paris. "She explained to me that she kept receiving calls from patients and that my gynecologist was going to remind me of." What she did the next day. "She immediately explained to me that I was not part of the most at risk as I am under 35 years old and that I have been on Luteran for less than five years," says Léa.
"The risk of meningioma was known"
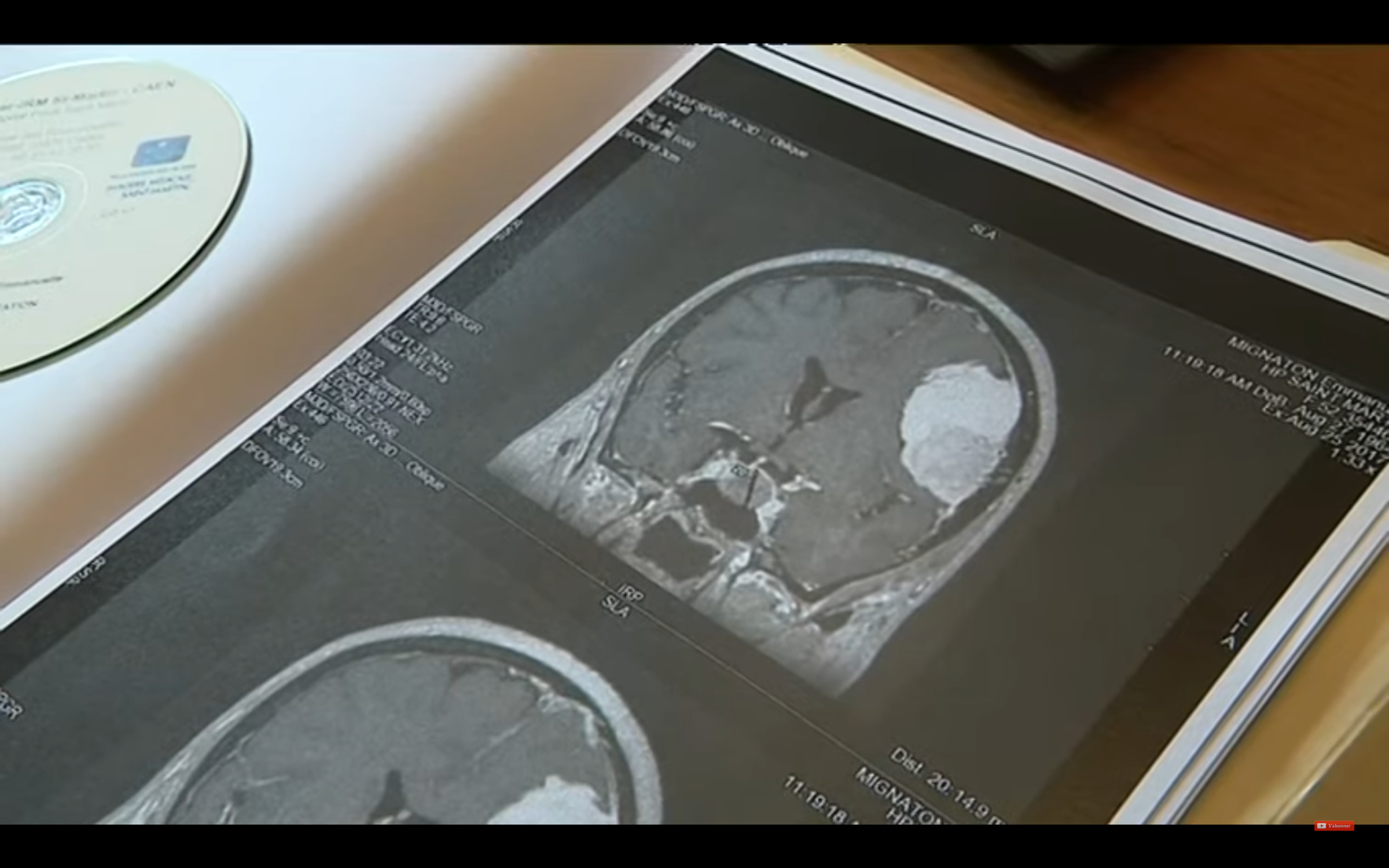
According to the epidemiological study at the origin of the alert, carried out by the Scientific Interest Grouping Ep-Phare constituted by the ANSM and the National Health Insurance Fund (CNAM), published in June 2020, a woman who takes Lotéran or Lotényl, for more than six months, has approximately 3.3 times more risk of developing meningiom . The risk increases according to the duration of the treatment and the age of the patient: under Lotényl, the risk is multiplied by 12.5 from five years of treatment and by 7 under Luteran after 3.5 years , according to the results of the study carried out on more than 3 million patients.
"I totally trust my gynecologist," continues the student. "She has always been very attentive and I think that if I had an emergency meeting, she would have told me." Even if the doctor was not very worried, Léa still decided to stop Lotéran, as a precaution and to reassure her relatives, very anxious. "I am waiting for my appointment in December, the result of my MRI (prescribed by his gynecologist, editor's note) before worrying, I don't want to rot my life."
On the same subject
• The contraceptive pill of the future will be organic, universal and without hormone
• Male contraception, can we pass the pill?
Same sound of bell for Julie, 22, suffering from endometriosis : "When I learned the news, inevitably I had a small climb of stress and then I told myself that I only took Ltéran for a year. In terms of risk, we were not on something very dangerous ”. Unlike Léa, Julie is aware of the on-risk of meningioma for a long time. Indeed, the ANSM has alerted since early 2019 to the risks of the Acetate molecules of Chlormadinone and Nomestrol acetate , present in the two pills and their generics. "The risk of meningioma was already known for Androcur (another progestogen," Grand-Frère "by Lutéran and Lotényl, editor's note) , and has been registered in the notice since 2011", specifies the ANSM in Neon. A report which was not made for Lutéran and Lotényl, however of the same family, until February 2019: "A letter was sent to healthcare professionals in order to inform them of the potential risk, and at this stage still undelicated, of meningioma (for these two drugs)", continues the ANSM. The link is now known and measured since the Epi-Phare study published in June. We understand better why none of the women we interviewed was warned by her gynecologist of this possible side effect.
"I never slept, for me, I was reached"
"In April 2019, I went around the pharmacies, because Lutéran was breaking everywhere and no one explained to me why," recalls Murielle, Julie's mother, who follows the same hormonal treatment. "I was baraticated a little, I was told" it had to change laboratory "or" there is no longer the molecule "". One day, a pharmacist finally informed him that his pill is withdrawn from the market and that she must contact her gynecologist. "To our appointment, she immediately said to me:" Yes, there is a risk of meningioma "".
A shock for the 51 -year -old woman, under Lutéran for 26 years: “ I was a zombie. I never slept, I was very anxious. For me, it was sure I was reached. "His new gynecologist for a year," who did not know his file well, "did not help. "She did not ask me the right questions, I think she minimized the risks, because I had no symptoms," regrets Murielle, who had to ask her a prescription for an MRI. Conversely, Julie believes that she was very well followed. "She exposed the benefit/risk on me on the phone and prescribed me an MRI," explains the student. She also told me that I could keep my pill if it suited me, because in my case, there was not much risks. The mother and daughter finally chose to interrupt their treatment even if their MRI did not show a tumor.
Luteran will no longer be marketed
"Developing meningioma is not systematic," reassures Isabelle Yoldjian, head of the gynecology pole of the National Health and Medicine Agency, in an interview for 20 minutes . "Meningioma is a tumor which is most often non-cancerous but it can be the cause of serious disorders that may require heavy and risk surgery," the ANSM specifies on the phone. "Women have to strive, ask their doctors questions, it's normal for them to worry. The ANSM has also communicated preliminary recommendations for patients and health professionals, such as consulting your doctor , requesting MRI - in the event of symptoms or if the patient is over 35 years old and has taken the pill for at least five years - and reassess the relevance of treatment .
Since the study, Luteran's marketing stop has been announced, even if it only concerns princeps, 30 % of the market, and not generics, specifies the ANSM. However, some patients may continue to find Lotéran. If there is no prizes recall, pharmacies will go to the flow of stocks.
After the stress of an over-risk of tumor comes to anxiety for a change of pill. And for good reason, finding a hormonal treatment that suits us is so complicated that when it is the case we would like to never part with it. "I don't know what my gynecologist will be able to prescribe me," says Léa who is afraid of a disruption. "Lotéran was a bit of my last recourse in terms of contraception".
*The first name has been changed
Online article: https://www.neonmag.fr/pilules-luteran-lutenyl-et-risque-de-tumeur-faut-sinquieter-561276.html
To see other publications click here