VISIO REUNION OF December 16, 2020
Temporary scientific committee
Lotényl / Lotéran and risk of meningioma
The members of the temporary scientific committee are:
Sébastien Froelich, president (neurosurgeon)
Geneviève Plu-Bureau (gynecologist)
Etienne Richer (gynecologist)
Jacques Young (endocrinologist)
Henry Dufour (neurosurgeon)
Martine Alt-Tebacher (CRPV pharmacologist)
Thierry Brillac (general practitioner)
Alain Weill (epiphare epidemiologist)
Sabine Trébaol (in charge of institutional relations of the Endofrance association)
Marianne Niosi (Executive Director of the National Confederation of Family Planning)
Angèle Mbarga (President of the Fibrome Info France association)
Axelle Ayad (representative of the Endomind association)
Emmanuelle Huet-Mignaton (president of the AMAVEA association)
Discussion around MRI
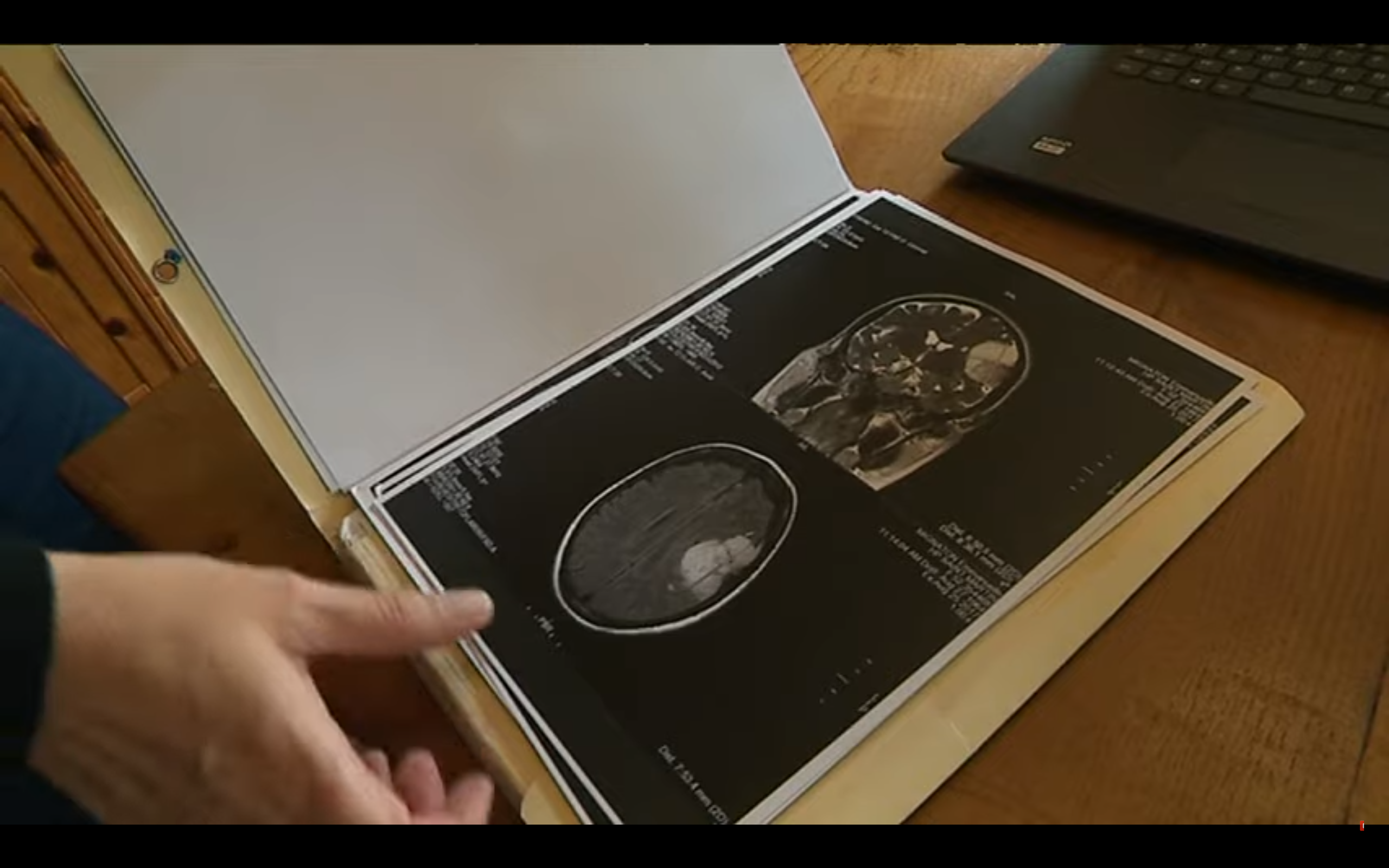
It is recalled that the reference technique to detect meningiomas is magnetic resonance imaging (MRI) with contrast product injection (Gadolinium). In case of contraindication to MRI, a scanner with injection can be offered.
The goal is to secure the treatment as much as possible, by passing MRI adequately.
- The cost of systematic MRI must be taken into account.
- Beware of women who have meningioma and who do not know it , and who often end up being operated on, because meningioma is discovered too late. The more meningioma is discovered early, the more heavy you have the least heavy care as possible.
- Likewise pregnancies are a frequent growth accelerator of meningiomas (natural progesterone that the body of the woman produces during pregnancy).
- Watch out for patients with Risk factors for meningiomas Who are:
- History of cranial radiotherapy in childhood
- Family context or history of neurofibromatosis (NF)
Whatever the age of the patient, an MRI is made after a year of treatment. And not on the initiative of treatment, because, Alain Weill, epidemiologist, said that half of the women stop treatment before 6 months, so it is not logical to do systematic MRI before starting treatment, while in 1 out of 2, it will be stopped quickly.
Too systematic MRIs update "small" problems, which would have gone unnoticed all life without this screening, and which will however be necessary to monitor. This situation generates additional anxiety.
On the other hand, systematic screening makes it possible to detect an aggressive tumor at an early stage, which can be held and heal more easily.
We can see that the meningiomas present before treatment do not regress the latter's stop. Hence the interest of detecting as much as possible of meningiomas, especially in young women (it has not been retained from "cut-off" of age to plan MRI, that is to say a different surveillance depending on the age of the patient).
Just as we do not know if a meningioma can appear after stopping treatment, be it androcur, luteran or lutenyl. If no MRI was made before the treatment initiative, or during treatment, it is impossible to assess.
The indications for these treatments
This meeting was also intended to prevent, in the future, the massive and abusive use of these 2 drugs that are Lutéran and Lotényl
The indications selected are (they will be detailed when the committee has validated them):
- Mainly those where the ovaries must be blocked, or for women awaiting surgical intervention (during these periods of Cavid, the wait can be long)
- Only when the therapeutic alternatives have failed or there is a contraindication to other treatments.
I personally specified what is written on the website of the Ministry of Health for the outset indications, that is this text:
Off-Amm prescriptions
In France, when they prescribe, doctors must respect the AMMs of pharmaceutical specialties. The non-compliance prescriptions for AMM are possible but supervised.
A pharmaceutical specialty can only be the subject of a non-compliance prescription to its MAM only in the absence of an appropriate drug alternative with an AMM or an ATU cohort and subject to the specialty for the purpose for the envisaged use of a temporary recommendation of use (RTU) drawn up by the ANSM or, in the absence, the prescriber judges essential, with regard to the data acquired from the science, Specialty to improve or stabilize the patient's clinical condition.
The doctor must relate to the prescription the mention "prescription outside the marketing authorization" and inform the patient of this out -of -AM use by presenting the advantages and risks of this prescription.
- Consult Articles L. 5121-12-1 and R. 5121-76-1 to R. 5121-76-9 of the Public Health Code
Isabelle Yoldjian indicated that the Outside AMM is to be proscribed in general, and that to withdraw a MA, European arbitration is needed. A product marketed in France must go through Europe (Ema- European Medecines Agency) to be modified.
Learned companies have the freedom to offer other additional clinical situations which they deem relevant via more detailed recommendations and under their responsibility (expert opinion or level of evidence based on literature).
Problem of meningiomas detected in a young woman, in desire for pregnancy, or in a woman whose treatment has a certain benefit
It was discussed the need to set up a multi-disciplinary consultation, with gynecologist/endocrinologist and neurosurgeon (and radiotherapist if necessary). This is a problem raised for over a year with Professor Froelich and which I also discussed with Gay Professor, president of the SFNC (French Society of Neurosurgery).
Is also discussed the need to make pre-conceptual consultations for women with meningiomas.
Risk of meningiomas with other progestins
According to Alain Weill, epidemiologist: if all the progestins could give meningiomas, given the number of women who take contraceptives, we have been able for a long time.
Meta-analyzes on the TSH (hormonal substitute treatment) reveals a small meningioma ride. TSH is less prescribed in France, due to the risk of breast cancer. These treatments are much less prescribed than 15 years ago, and over a shorter period.
There are no meta-analyzes on microprogestatives and the risk of meningiomas.
(Precision of the association: The request for an epidemiological study was made in the meeting of the CSP Pharmacovigilance of December 15, 2020, on the initiative of the Amavea association).
Information to professionals and patients
Given the importance of information to deliver to professionals and patients concerning the risk of Méningioma poru these 2 treatments (as was for Androcur in 2019), it was decided that a meeting only dedicated to this theme would be scheduled in January 2021 in order to discuss in particular the establishment of patient documents (e.g. treatment certificate).
Emmanuelle Huet-Mignaton
To go further on AMM for a drug
Answer here on an official website, VIE-PUBLIE.FR, an information site produced by the DILA (Directorate of Legal and Administrative Information), attached to the services of the Prime Minister.
Drug legislation in France is very strict for health security reasons. New drugs (as well as vaccines) go through a long process in order to obtain a marketing authorization (MA). Back in nine questions about this market to market.
What is marketing authorization (AMM)?
The pharmaceutical company which wishes to market a new drug must submit an authorization to market to market (AMM) to the competent, national (National Agency for Medicines and Health Safety Agency - ANSM) or European (the European Agency for the Evaluation of European Medicines Agency - EMA).
This file includes the results of the numerous research and clinical and preclinical research works. He must guarantee:
- pharmaceutical quality (composition, stability, sterility, large -scale reproducibility, etc.);
- employment safety (undesirable effects, precautions for use, etc.);
- the effectiveness of the drug (test data concerning the experimental pharmacological efficiency of the product).
The AMM is issued, depending on the procedure, either by the ANSM or by the EMA. She is accompanied:
- the summary of the characteristics of the product (RCP) (name, composition, contraindications, undesirable effects, etc.);
- of the manual for the patient;
- labeling.
ATU, RTU, AMM: What differences?
A temporary authorization for use (ATU) (new window) , issued by the ANSM, allows access to drugs that have not yet, or not, AMM. They are, in principle, intended for patients with rare or serious diseases that do not have another appropriate and available treatment. Their efficiency and employment of employment are presumed in the state of scientific knowledge and the implementation of treatment cannot be deferred. The ATU is issued for a limited period.
The temporary recommendation for use (RTU) (new window) concerns drugs that already have a MA. RTU authorizes another therapeutic use. The ANSM issues this “excluding MA” recommendation for a maximum duration of three years.
What is the role of the ANSM in the medication course?
The ANSM was created in 2012 after the Mediator® crisis. It has administrative police powers in terms of health products for humans and scientific expertise. Its main mission is to assess the benefits and risks of health products and to guarantee their safety throughout their life cycle:
- It assesses the quality and safety of drugs, monitors their undesirable effects;
- It inspects establishments that produce drugs or carry out clinical trials;
- It authorizes drugs by delivering MA (whether it can also suspend or remove).
- It allows clinical trials;
- It releases lots of vaccines and blood derived from blood;
- It authorizes and controls medical devices.
What are the tests carried out upstream of clinical tests?
When the most promising compounds against a specific disease were selected during a first step in the research laboratory, they are subject to so-called pre-clinical trials. The purpose of these tests is to understand the mode of action of the compounds, to assess their effectiveness and their toxicity. Possible side effects are sought. The tests are carried out mainly on the animal.
Note
Animal experimentation for drugs is considered essential to study the interactions between the different structures of an entire organism and assess side effects. This experiment is based on the 3R rule: replacement (replace the animal experiences as much as possible), reduction (limit the number of trials and animals), refinement (use techniques with the least possible constraints for the animal).
What are the four phases of a clinical trial?
The first clinical trials on humans start after obtaining satisfactory results during the pre-clinical phase. They are strictly supervised by law and can only start after favorable advice from the Person Protection Committee and authorization from the ANSM. The four phases of a test are as follows:
- Phase I : tests carried out on a small group of non -sick volunteers (twenty) in order to verify the tolerable doses of the drug in humans and its future in the body;
- Phase II : Tests carried out on a few hundred participants with targeted disease. The optimal product dosage is determined and the side effects observed;
- Phase III : trials carried out on a large population of volunteers with targeted disease (up to several thousand), on several sites. It is a question of comparing the effectiveness of the drug in development with that of an already existing treatment and/or that of a placebo, that is to say a treatment without pharmacological activity. This phase, which lasts several years, aims to answer the question of the benefit of the drug for the patient as well as to identify the potential risks. We are talking about a profit/risk ratio. In parallel, an industrial development phase takes place which determines the mode of administration and packaging of the active ingredient. It is at the end of phase III that the AMM file constituted can be subject to the ANSM or to the European Health Authorities;
- Phase IV : Large -term surveillance phase tests and in the long term, after obtaining the MA. We are talking about pharmacovigilance .
What do we mean by "benefit/risk ratio"?
The essential objective of a medication is to benefit the patient, to cure him or to alleviate his symptoms. But taking a medication is not without risk. The MA should only be given if the benefit/risk ratio is deemed favorable. For some very severe diseases such as cancer, this report is evaluated differently and it will be accepted that there are risks such as hair loss or nausea. In other circumstances, these risks will be deemed unbearable.
What is happening after the marketing of the drug?
Once a drug is put on the market, its impact is studied on a very large population in order to prevent and follow its undesirable effects. It is pharmacovigilance.
Born in 1974, the French pharmacovigilance system is based on the spontaneous notification of health professionals (doctors and pharmacists essentially who signal the side effects of drugs). Health scandals, in particular that of the Mediator® medication, have questioned this device, its independence and the competence of the actors responsible for the control of drugs, from health agencies to prescribing doctors. The integrity of the pharmaceutical industry was also interviewed.
It is in this context that the law of December 29, 2011 was adopted relating to the strengthening of the health security of drugs and health products (new window) . It aims to clarify the roles of the different actors and extends the circle of notifiers to patients and patient associations. The law of January 26, 2016 of modernization of the health system initiated a national reform of health vigilances.
Why can a drug be removed from the market?
Depending on the severity of the side effects and the benefit/risk ratio noted or in the event that the conditions for granting the AMM is not respected, the ANSM may suspend, modify or withdraw a market for market.
The decision to suspend an AMM intervenes in particular when it appears that the drug is harmful under normal conditions of employment or that the therapeutic effect is lacking or that the drug does not have the quantitative and qualitative composition declared.
In some cases where the undesirable effects are important but the drug remains very useful, its dissemination and its administration can then be limited, for example, in the hospital environment.
ANSM meeting of December 16, 2020 - CST "Lotényl / Lotéran and risk of meningioma"
Find our other articles here