Our systematic review with meta-analysis on the risk of intracranial meningiomas associated with progestin drugs, conducted in partnership with GHU PARIS, neurosurgery department of Professor Johan Pallud.
By analyzing 78 international studies, we show:
– a major increased risk with cyproterone acetate (estimated at 12.4)
– an increased risk with medroxyprogesterone acetate
– signals for other macroprogestins (chlormadinone acetate, nomegestrol acetate, promegestone, medrogestone, desogestrel)
– An increased risk of malignant meningiomas for certain molecules
– A predominance of locations at the base of the skull
– A possible but not systematic regression of the size of the meningioma after stopping treatment.
The level of evidence varies according to the molecules, but these data reinforce the need to clearly inform patients, to limit prescriptions (strictly necessary indication, minimum dose, minimum duration) and to ensure appropriate monitoring in case of prolonged exposure .
Read the study here: https://www.thelancet.com/journals/eclinm/article/PIIS2589-5370(26)00038-6/fulltext
Thanks to Benoit Hudelist and the entire neurosurgery team at GHU Paris Psychiatry and Neuroscience – Sainte-Anne for this collaborative work.
Summary
Background. Meningiomas are the most common primary intracranial tumors in adults. A potential link between progestin exposure and the occurrence of intracranial meningiomas is a cause for concern. We evaluated the association between different progestins and the risk of intracranial meningioma, as well as secondary outcomes (malignancy, location, regression).
Methods. We conducted a systematic review and meta-analysis of epidemiological studies (English/French) reporting an association between progestin exposure and intracranial meningioma. The PubMed/MEDLINE, Embase, Cochrane Library, and EPI-PHARE databases were searched from their inception to November 1, 2025, supplemented by pharmacovigilance reports and a reference search. Excluded were non-original studies, abstracts without full text, and studies without eligible exposure or without relevant outcome measures. The risk of bias was assessed using the Newcastle-Ottawa Scale, and the certainty of the evidence was assessed using GRADE. Random-effects models were used; heterogeneity was estimated using I²; a narrative synthesis completed the analysis.
Results. Of 542 references screened, 78 studies were included in the review and 14 high-quality observational studies in the meta-analysis. Cyproterone acetate (CPA) was strongly associated with an increased risk of meningioma (5 studies; 1047 exposed cases; pooled OR 12.36 (95% CI 7.47–20.45); I² 73.8%; moderate GRADE). Medroxyprogesterone acetate depot was also associated (6 studies; 842 exposed cases; OR 2.68 (1.72–4.19); I² 92.7%; low GRADE). Signals of increased risk were reported for chlormadinone acetate, nomegestrol acetate, promegestone, medrogestone, and desogestrel. Regression after discontinuation was described for CPA and nomegestrol. The tumors were mostly located at the anterior/mid-skull base, and malignant forms appeared to be more frequent with CPA, chlormadinone, and nomegestrol.
Interpretation. Despite limitations related to the observational nature of the study, residual confounding factors, heterogeneity, and imprecision for certain exposures, some progestins, particularly high-dose macroprogestins, may increase the risk of intracranial meningioma. Clear patient education and clinical, and radiological if indicated, follow-up are essential.
Data available prior to this study
Before undertaking this study, we searched PubMed/MEDLINE, Embase, and the Cochrane Database of Systematic Reviews from the databases' inception to November 1, 2025 (without language restrictions) to identify prior systematic reviews and meta-analyses on systemic progestins and the risk of intracranial meningioma. We also examined the reference lists of eligible reviews and key pharmacovigilance/regulatory documents (e.g., EMA, ANSM/EPI-PHARE). The search terms combined "meningioma" with "progestin/progestin." We included evidence syntheses with explicit eligibility criteria and a structured risk of bias assessment, and excluded narrative reviews and case reports/series. We identified a previous systematic review/meta-analysis focused on cyproterone acetate, reporting an imprecise combined estimate (RR: 3.78, 95% CI: 0.31–46.39) and judged to have a moderate to high risk of bias; certainty was low. No previous reviews comprehensively evaluated other progestins or outcomes such as tumor regression, risk of malignancy, or skull base predilection.
Added value of this study
This systematic review and meta-analysis synthesizes the largest contemporary evidence base on macro- and microprogestins in diverse populations. It identifies specific agents associated with an increased risk of meningioma and confirms a class effect for macroprogestins. It also highlights desogestrel as a microprogestin associated with meningioma during prolonged use. Beyond incidence, we document consistent radiological regression or stabilization after treatment discontinuation, quantify the elevated risk of malignant meningioma (WHO grade 3) for certain agents, and delineate a predilection for anterior and midline skull base locations—findings directly relevant to screening and management. Overall, this study consolidates current knowledge and provides a solid foundation to guide clinical practice, policy, and future research.
Implications of all available data
Given the widespread and often prolonged use of progestins for contraceptive purposes, to treat endometriosis, as part of menopausal therapy, and to suppress androgens, clinicians should routinely document progestin exposure in patients with or suspected of having meningioma, consider stopping high-risk agents where possible, and consider MRI follow-up in accordance with national guidelines and individual risk profiles, particularly in women with prolonged exposure to high doses of macroprogestins or with or suspected of having meningioma. However, interpretation must take into account some important limitations: most of the available data are observational, exposure definitions and dose thresholds vary across studies, heterogeneity is significant for some pooled estimates, and residual confounding factors and surveillance (detection) bias can inflate risk estimates, particularly in settings where MRI screening is structured. Evidence for several agents remains limited and imprecise, and its generalizability to men and transgender populations is uncertain due to incomplete sex/gender reporting. Prescribers and regulators should integrate agent-specific risk profiles into an overall class-effect framework for macroprogestins and reassess recommendations for microprogestins as higher-quality comparative studies become available.
Introduction
Meningiomas are the most common primary tumors of the central nervous system in adults, accounting for approximately 40% of cases, with an incidence of 9.5 per 100,000 person-years. Most meningiomas are benign and slow-growing. They may be discovered incidentally or present with neurological symptoms such as seizures, focal deficits, neurocognitive impairment, or headaches. Their incidence is higher in females, with a male-to-female ratio of 2:1 after puberty, increasing to 3:1 during the reproductive years. Progesterone plays an important role in the pathophysiology of meningiomas: an increase in meningioma size has been reported during pregnancy due to elevated progesterone levels, and studies have documented a prevalence of 38% to 88% of progesterone receptors in meningioma cells. Reports of intracranial meningiomas in patients treated with high doses of cyproterone acetate (CPA) and other potent progestins have raised concerns about a potential causal link and prompted specially designed epidemiological studies. Progestins are prescribed worldwide for both approved indications (e.g., contraception, endometriosis, and transgender hormone therapy) and off-label uses (e.g., acne and female pattern baldness). Medroxyprogesterone acetate (DMPA) is one of the most widely used injectable contraceptives globally, particularly in large-scale family planning programs, resulting in prolonged exposure for millions of women. Large-scale cohort studies have explored the link between progestin exposure and the risk of meningioma, providing significant data with socioeconomic implications, given the widespread use of progestins. Because meningiomas are slow-growing tumors, any causal effect of progestins is expected to manifest primarily after long-term or high cumulative exposure, while short-term treatments are unlikely to have a significant impact on risk. This systematic review and meta-analysis summarizes current knowledge on the relationship between meningiomas and progestins, with a particular focus on clinical implications, paying special attention to dose, route of administration, duration of exposure, and clinical implications for patient management.
Methods,
research strategy and selection criteria
This systematic review was conducted and reported in accordance with the MOOSE guidelines and the PRISMA statement, following recent methodological recommendations for meta-analyses in neurosurgery. The study was reviewed and approved by an institutional ethics committee (IRB00011687, IRB #1: 2024/53).
The EPPI Reviewer software (v.6.15) was used for study selection and screening. From database creation until November 1, 2025, we searched the US National Library of Medicine (PubMed/MEDLINE), Embase (OVID), the Cochrane Library, and the EPI-PHARE database to identify all epidemiological studies examining associations between intracranial meningiomas and systemic progestin exposure. Duplicate records were identified and removed. Study titles and abstracts were reviewed against inclusion criteria. Full-text studies were imported and assessed for inclusion. In addition, we used backward snowballing and reviewed pharmacovigilance reports and national/international regulatory guidelines (including those of the EMA and national agencies). The first author (BH) reviewed titles/abstracts; The lead author (JP) reviewed the full texts and validated the exclusions based on the abstracts; disagreements were resolved by consensus or, if necessary, by a third reviewer (AR).
Publications were eligible if they met the following criteria: (1) full-length articles written in English or French; (2) reporting an association between progestin exposure and meningiomas; and (3) intracranial location of the meningioma. Systemic progestins administered orally, by injection, or intrauterinely were eligible; purely local or topical preparations without significant systemic absorption were excluded. Only epidemiological studies (cross-sectional, case-control, cohort) using evidence-based methods were included in the meta-analysis. Descriptive cross-sectional reports, pharmacovigilance case series, and experimental studies were summarized narratively where relevant but were not pooled.
Using a standardized form, we extracted data on study characteristics (country, year, design, and number of cases), sex (as reported in the original articles), and imaging characteristics (tumor volume, time course, and location). For progestins, we collected characteristics (type, dose, duration, and risk of meningiomas) and route of administration (oral, injectable, or intrauterine). We used the Newcastle-Ottawa Scale (NOS) to assess the methodological quality of non-randomized studies. This scale evaluates quality in three areas: (1) study selection (4 points), (2) comparability of groups (2 points), and (3) exposure or verification of results (3 points). Studies scoring 7 points or higher were classified as high quality. Two reviewers (BH and JP) independently applied the NOS scale and resolved discrepancies by consulting a senior author (RA) when necessary. The certainty of the evidence was assessed using the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach. GRADE assessments were performed at the level of each molecule-exposure contrast included in the quantitative synthesis, starting from a "low" certainty for observational data and downgrading the risks of bias, inconsistency, imprecision or suspected publication bias, where applicable; for exposures with significant and consistent effects and a plausible dose-response gradient, we considered raising them by one level.
When multiple studies assessed similar exposures based on potentially overlapping populations and time periods, we prioritized cohort studies over case-control studies, favoring those with larger samples and more robust dose-response analyses. This strategy aimed to minimize the risk of double-counting individuals and to preserve the independence of the effect estimates included in the meta-analysis.
Data analysis
We performed random-effects meta-analyses on the logarithmic scale of relative risks using generic inverse-variance weighting and a restricted maximum likelihood estimator for τ². Effect estimates were log-transformed before pooling and then back-transformed for interpretation. Heterogeneity was summarized by I² and Tau.². Forest plots display pooled relative risks with a 95% confidence interval. We interpreted I² as low (<30%), moderate (30–60%), substantial (60–90%), and considerable (>90%). Where heterogeneity was substantial, we also reported 95% prediction intervals and performed leave-one-out influence analyses. Quantitative synthesis was limited to molecule-specific contrasts with at least two comparable studies. All analyses were performed using R (v4.0.5) and the meta and metafor packages.
We predefined the odds ratio (OR) as a pooling measure and performed meta-analyses on the log-OR scale using the generic inverse variance method. When only the relative risk (RR) or the human risk ratio (HR) were available, we used the adjusted estimates reported on the logarithmic scale and treated them as approximations of the ORs, given the low absolute incidence of intracranial meningiomas.
Role of the funding source
This study received no funding. The authors had full access to all data included in the study. BH and JP assumed final responsibility for the decision to submit the article for publication.
Results
The PRISMA flow diagram is shown in Figure 1.
Initially, 542 studies were identified: eight duplicates were removed, 436 were excluded based on title and abstract, 12 were not retrieved, and eight were excluded based on full text. Ultimately, 78 studies were included in the final review . Of these, 14 studies were deemed eligible for inclusion in the meta-analysis (presented in Table 1), 5 were cohort studies, and 9 were case-control studies. All 14 studies were rated as high quality according to the NOS rating system; however, even studies rated as "high quality" according to the NOS remained subject to residual confounding factors and, in some registry-based analyses, to misclassification of outcomes. According to the GRADE system, the certainty of the evidence was moderate for high-dose CPA and low for DMPA, and low to very low for other progestins (reflecting observational design, residual confounders, heterogeneity, and imprecision across multiple exposures).
Characteristics of the included studies.
CPA: cyproterone acetate; NOMAC: nomegestrol acetate; CMA: chlormadinone acetate; DMPA: injectable medroxyprogesterone acetate.
The classification of synthetic progestins, their modes of action, their indications and their dosages are summarized in Table 2.
Table 2
Summary of evidence on the risk of intracranial meningioma associated with progestins.
*Restricted/monitored use in several countries due to the risk of meningioma (class/molecule specific).
†Contexts vary by country and may be off-label.
%Presented as combined OR (CPA and DMPA) or OR.
∗∗GRADE certainty ratings were assigned to exposures for which sufficient comparative epidemiological data existed; for other exposures, the data were insufficient to support a GRADE assessment.
HRT: hormone replacement therapy; PMS: premenstrual syndrome; UA: abnormal uterine bleeding. [BH1]
Risk of meningioma
Cyproterone acetate (CPA)
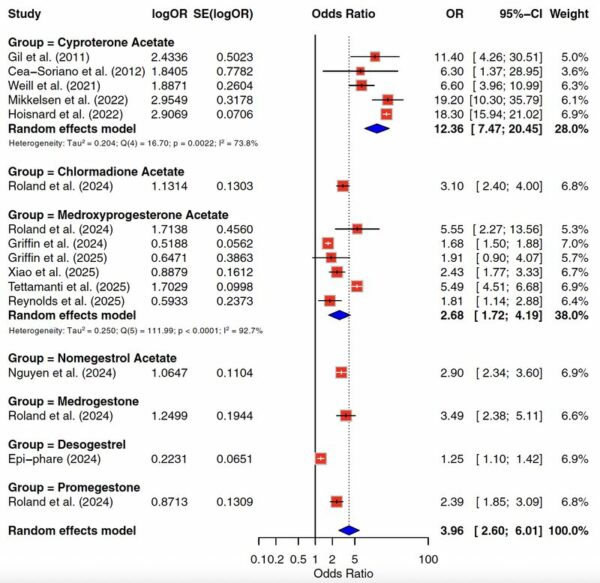
CPA was the first progestin identified in 2007 as potentially associated with meningiomas. Since then, case reports and cohort studies have documented the occurrence of meningiomas in the context of CPA exposure. All studies converge on a clear association between high doses or high accumulation of CPA. Gil et al. found in 2011 an increased risk of meningioma in people using high doses of CPA (aRR: 11.4, 95% CI: 4.3–30.8), with 4 cases out of 2,474 people exposed to CPA compared to those not exposed to CPA. Cea-Soriano et al. identified in 2012 an increased risk of meningioma associated with exposure to high doses of CPA (OR: 6.30, 95% CI: 1.4–28.9; 3 cases), but not with low doses. Exposure to high doses of CPA was almost exclusively male, while exposure to low doses of CPA in combined oral contraceptives was more common in women. A large French cohort study by Weill et al. in 2021 identified a risk of meningioma in patients exposed to CPA (HR: 6.6, 95% CI: 4.0–11.1; 69 cases). They observed a dose-response relationship with a higher risk for high cumulative doses of CPA (>60 g; HR: 21.7, 95% CI: 10.8–43.5; 15 cases). Notably, one year after cessation of CPA exposure, the risk of meningioma remained higher in the exposed group than in the unexposed group (HR: 1.8, 95% CI: 1.0–3.2; 34 cases). A subsequent French study by Hoisnard et al. in 2022 confirmed the risk of meningioma associated with CPA exposure (OR: 18.3, 16.0-21.1; 961 cases). They observed the strongest association with CPA exposure > 1 year (OR: 22.7, 95% CI: 19.5-26.6; 931 cases). A Danish cohort study conducted by Mikkelsen et al.60 in 2022 confirmed the dose-dependent association between CPA exposure and the risk of meningioma (cumulative CPA exposure of 0.1 to 10 g, HR: 7.0, 95% CI: 3.1-15.6, 6 cases/cumulative exposure > 10 g, HR: 19.2, 95% CI: 10.3-35.8, 10 cases). A previous meta-analysis conducted by Lee et al. attempted to pool data on progestins and meningiomas, but it contained several numerical inaccuracies (e.g., incorrect sample sizes in the main registry studies and inconsistent treatment of person-time) and reported a non-significant association between CPA use and the risk of meningioma (RR: 3.78, 95% CI: 0.31–46.39). Given these methodological problems, we did not rely on their pooled estimates and instead presented an independent synthesis based on a new extraction and analysis of the original studies. Several studies focused solely on low doses of CPA, and none found a link with meningioma, even with long-term use. The recent report from the EPI-PHARE Scientific Interest Group of the French National Agency for Medicines and Health Products Safety, authored by Roland et al. A 2023 study confirmed the significant association between CPA exposure and the risk of meningioma (OR: 19.21, 95% CI: 16.61–22.22; 891 cases).
After converting the HR/RR to OR and restricting the analysis to exposure to high or high cumulative doses of CPA, the combined OR was 12.36 (95% CI: 7.47–20.45), with significant heterogeneity (I² = 73.8%, τ² = 0.204; n = 5 studies, 1047 cases). Leave-one-out analyses did not change the direction. Studies evaluating the risk of meningioma associated with CPA use are summarized in Figure 2 and Table 3.
Table 3
Summary of combined relative risks for progestin exposure with formal meta-analyses.
Participants providing data refer to the number of meningioma cases (events) among exposed participants contributing to the combined estimate; total cohort sizes were not consistently reported across all studies.
Chlormadinone acetate (CMA)
The potential link between CMA and meningiomas was first reported by Shimizu et al. in 2008. In 2020, Roux et al. reported another case of meningioma volume reduction after CMA discontinuation, further suggesting a possible link. In 2021–2022, Malaize et al., Graillon et al., Devalckeneer et al., and Samoyeau et al. reported stabilization or regression of meningiomas after CMA discontinuation. Prior to 2022, Grandi et al. cautioned against drawing any definitive conclusions due to the limited amount of available data. An epidemiological study conducted by the EPI-PHARE Scientific Interest Group by Hoisnard et al. in 2022 identified a significant risk of meningiomas with CMA exposure (OR: 4.7, 95% CI: 4.5–5.3; 683 cases). A 2021 EPI-PHARE SNDS (French National Health Data System) cohort report identified for the first time an increased risk of meningioma with prolonged exposure to high doses of CMA, which was subsequently confirmed in the peer-reviewed update by Roland et al. in 2024 (age-adjusted RR: 3.1, 95% CI: 2.4–4.0; 164 cases). In the highest-exposure group (cumulative dose) (>8.64 g), the age-adjusted RR of developing a meningioma was 6.9 (95% CI: 5.1–9.2; 86 cases) compared to the control group, suggesting a dose-response effect. The most recent report from the EPI-PHARE scientific interest group confirmed the significant association between exposure to CMA and the risk of meningiomas (OR: 3.87, 95% CI: 3.48-4.30; 628 cases).
Given the limited number and heterogeneity of the designs, we did not perform a pooled meta-analysis for CMA and provide a narrative synthesis (summarized in Figure 2).
Medroxyprogesterone acetate (MPA)
The potential association between MPA and meningiomas was first evaluated by Korhonen et al. in 2012 and did not identify an increased risk. The study's limitations included the inclusion of all progestins in the same group and the restriction to exposure to postmenopausal hormone replacement therapy only. Abou-Al-Shaar et al. were the first to suggest, in 2023, an association between MPA use and the risk of meningioma. Pourhadi et al. conducted an epidemiological study in Denmark in 2023 on postmenopausal hormone therapy and meningiomas without specifically analyzing MPA. They reported a higher risk in both the estrogen-progestin group (HR: 1.21, 95% CI: 1.06-1.37; 423 cases) and the progestin-only group (HR: 1.28, 95% CI: 1.05-1.54; 143 cases), suggesting a potential association between progestin-containing menopausal hormone therapy, largely based on MPA in this setting, and meningioma.
A case-control study based on the US registry by Griffin et al. in 2024 suggested that oral exposure to MPA was not associated with an increased risk of meningioma, unlike exposure to DMPA (OR: 1.68, 95% CI: 1.50-1.87; 480 cases), and that the risk increased with the duration of exposure (exposure ≤ 1 year, OR: 1.23, 95% CI: 1.10-1.38; 457 cases, and exposure > 3 years, OR: 2.50, 95% CI: 2.06-3.04; 170 cases). The 2024 report from the EPI-PHARE scientific interest group reported a significant association between DMPA exposure and meningiomas in France (OR: 5.55, 95% CI: 2.27–13.56; 9 cases). More recently, in 2025, several large-scale population-based studies specifically focused on DMPA confirmed this signal: Xiao et al. found, in 2025, an increased risk of meningioma among people using DMPA (RR: 2.43, 95% CI: 1.77–3.33; 131 cases), Reynolds et al. reported in a nested case-control study a high risk associated with "anytime use" of DMPA (OR: 1.81, 95% CI: 1.14-2.89; 29 cases), with significantly higher estimates for prolonged exposure and a low or no additional risk for short-term use. Tettamanti et al. observed, in a nationwide case-control study in Sweden, a strong association between DMPA and meningioma (RR: 5.49, 95% CI: 4.51-6.67; 186 cases). Furthermore, a more recent US case-control study by Griffin et al.76 using active and inactive hormonal comparators reported a non-significant but consistent increased risk associated with prior DMPA exposure (OR: 1.91, 95% CI: 0.99–4.50; 7 cases), reinforcing the hypothesis of a possible class effect of DMPA. Overall, these converging data indicate that the increased risk is largely due to prolonged or repeated DMPA exposure, while oral MPA and short-term treatments do not show a consistent association with meningioma.
The combined OR, limited to DMPA, was 2.68 (95% CI: 1.72–4.19), with significant heterogeneity (I² = 92.7%, τ² = 0.25; n = 6 studies, 842 exposed cases). Leave-one-out analyses did not change the direction. Studies evaluating the risk of meningioma associated with DMPA use are summarized in Figure 2 and Table 3.
In contrast, studies evaluating oral MPA, primarily in the context of menopausal hormone therapy or progestin-only oral therapy, have generally reported no or only a modest increase in the risk of meningioma. Overall, these results confirm that any potential risk associated with oral MPA is likely significantly lower than that observed with long-acting DMPA and may be limited to long-term, continuous use in specific populations.
Nomegestrol acetate (NOMAC)
The potential link between NOMAC and meningioma was first reported by Gruber et al. in 2004. In 2011, Gruber et al. reported four cases of patients in whom a meningioma was diagnosed during NOMAC treatment, and none showed signs of recurrence after surgery and discontinuation of NOMAC. Seven other case reports described similar cases of meningioma in patients treated with NOMAC, and numerous studies have been published on patients with meningioma exposed to NOMAC. Between 2021 and 2022, Samarut et al., Malaize et al., Graillon et al., Devalckeneer et al., and Samoyeau et al. suggested a link between NOMAC use and meningioma. However, the small sample sizes (ranging from 1 to 12 patients) did not allow for a definitive conclusion.
An epidemiological study conducted by the EPI-PHARE scientific interest group identified a significant risk of developing meningioma in patients exposed to NOMAC (OR: 4.7, 95% CI: 4.3–5.1; 969 cases). EPI-PHARE first reported an increased risk of meningioma with prolonged/high-dose NOMAC exposure in a 2021 SNDS study, which was subsequently confirmed and expanded upon in an updated 2024 analysis (age-adjusted RR: 2.9, 95% CI: 2.4–3.7; 171 cases), with a marked dose-response gradient (RR 12.0 for cumulative exposure > 6 g). The latest report from the EPI-PHARE scientific interest group published in 2024 reported a significant association between NOMAC exposure and meningiomas (OR: 4.93, 95% CI: 4.50-5.41; 925 cases).
Given the limited number and heterogeneity of the designs, we did not perform a pooled meta-analysis and provide a narrative synthesis (summarized in Figure 2).
Promestone
Abijaoude et al. reported a single case of osteomeningioma occurring during promegestone treatment in 2021. Apra et al. in 2020 and Graillon et al. in 2021 reported case series where meningiomas were observed concurrently with promegestone treatment, suggesting a potential association. The recent report from the EPI-PHARE Scientific Interest Group identified a significant risk of developing a meningioma in patients exposed to promegestone (OR: 2.39, 95% CI: 1.85–3.09; 83 cases). Prolonged use of promegestone for ≥ 1 year was associated with a higher risk (OR: 2.74, 95% CI: 2.04–3.67; 66 cases).
Given the limited number and heterogeneity of the designs, we did not perform a pooled meta-analysis and provide a narrative synthesis (summarized in Figure 2).
Medrogestone:
The potential association between medrogestone and meningiomas was first suggested by Apra et al. in 2020. The EPI-PHARE Scientific Interest Group report by Roland et al. identified a significant risk of developing a meningioma in patients exposed to medrogestone (OR: 3.49, 95% CI: 2.38–5.10; 42 cases). Prolonged use of medrogestone for ≥ 1 year was associated with a higher risk (OR: 4.08, 95% CI: 2.72–6.10; 40 cases).
Given the limited number and heterogeneity of the studies, we did not perform a pooled meta-analysis and provide a narrative synthesis (summarized in Figure 2).
Desogestrel:
The link between desogestrel and meningiomas was first investigated in the recent report by the scientific interest group EPI-PHARE. This group identified a significant risk of developing a meningioma in patients exposed to desogestrel (OR: 1.25, 95% CI: 1.10–1.42; 287 cases). The risk appears after five years of desogestrel use (OR: 1.70, 95% CI: 1.39–2.08; 115 cases) and increases with the duration of desogestrel exposure (5–6 years of use, OR: 1.51, 95% CI: 1.17–1.94; 71 cases, and ≥ 7 years of use, OR: 2.09, 95% CI: 1.51–2.90; 44 cases). If desogestrel had been discontinued for more than one year, the risk of developing a meningioma disappeared (OR: 0.83, 95% CI: 0.63-1.09; 58 cases). Building on these data, Roland et al. published a large national case-control study in 2025, based on the same cohort, and demonstrated that prolonged use (≥ 5 years) of 75 μg of desogestrel was associated with an increased risk of intracranial meningioma (OR: 1.70, 95% CI: 1.39-2.08, 115 cases), with a stronger association for use ≥ 7 years (OR: 2.09, 95% CI: 1.51-2.90, 44 cases).
Given the limited number and heterogeneity of the designs, we did not perform a pooled meta-analysis and provide a narrative synthesis (summarized in Figure 2).
Other progestins
Several studies have explored the association between other progestins (including dydrogesterone, levonorgestrel, norethisterone, progesterone, hydroxyprogesterone, spironolactone and dienogest) and meningiomas.
None of them identified a clear link. The EPI-PHARE scientific interest group studied the use of widely prescribed progestins in France between 2009 and 2018. It found no significant association between an increased risk of surgery for intracranial meningioma and exposure to oral or intravaginal progesterone (OR: 0.88 (95% CI: 0.78-0.99); 329 cases), percutaneous progesterone (OR: 1.11 (95% CI: 0.89-1.40); 90 cases), dydrogesterone (OR: 0.96 (95% CI: 0.81-1.14); 156 cases), spironolactone (OR: 0.95 (95% CI: 0.84-1.09); 264 cases), dienogest (OR: 1.48 (95% CI: 0.41–5.35; 3 cases), 52 mg levonorgestrel intrauterine system (OR: 0.94 (95% CI: 0.86–1.04); 566 cases), and 13.5 mg levonorgestrel intrauterine system (OR: 1.39 (95% CI: 0.70–2.77); 10 cases). No cases of exposure to hydroxyprogesterone were found. However, the EPI-PHARE Scientific Interest Group emphasized that no conclusions could be drawn regarding dienogest or hydroxyprogesterone due to the small number of people receiving these medications.
Given the limited number and heterogeneity of the designs, we did not perform a pooled meta-analysis and provide a narrative synthesis (summarized in Figure 3).
Fig. 3 Progestins not associated with an increased risk of meningiomas.
Risk of malignant meningioma
Grade 3 malignant meningiomas according to the WHO classification are rare, representing 1 to 2% of cases.<sup>1</sup> As reported by Roux et al. in 2022, cases of malignant meningiomas have been observed in association with progestins, suggesting a potentially higher risk associated with these drugs. The EPI-PHARE<sup>3</sup> Scientific Interest Group<sup>⁹</sup> demonstrated that progestin exposure was associated with a significantly increased risk of malignant meningiomas in women for CPA (OR: 23.7, 95% CI: 8.1–69.8; 20 cases), NOMAC (OR: 4.9, 95% CI: 2.2–10.8; 13 cases), and CMA (OR: 2.8, 95% CI: 1.3–5.9; 11 cases). These findings were confirmed by Roland et al. In 2024, a significant increase in the risk of malignant meningiomas was reported for CPA (OR: 22.5, 95% CI: 7.61–66.48; 18 cases), NOMAC (OR: 4.95, 95% CI: 2.29–10.68; 13 cases), and CMA (OR: 5.78, 95% CI: 2.39–14.00; 11 cases). No malignant meningiomas were observed in cases exposed to medrogestone, medroxyprogesterone acetate, or promegestone.
Meningioma growth
The effects of drug withdrawal on the spontaneous growth of untreated meningiomas have been reported in 16 studies for CPA (ranging from 1 to 188 cases), in 10 studies for NOMAC (ranging from 1 to 6 cases), in seven studies for CMA (one case per study), in one study for MPA70 (10 cases), and in two studies for promegestone (one case per study). No studies were identified for other progestins.
For CPA, stopping treatment leads to a significant reduction in tumor size on MRI: regression in 36 to 92% of cases, stability in 8 to 85.2% of cases, and continued growth in 0 to 29% of cases.42 Most studies focused on various progestins and examined volume changes in all patients exposed to progestins, making it difficult to draw conclusions for each one: regression in 0 to 12.5% of cases, stability in 60.0 to 62.5% of cases, and continued growth in 25.0 to 40.0% of cases for NOMAC³⁸; no regression, stability in 66.7% of cases, and continued growth in 33.3% of cases for CMA.38 Graillon et al. combined data from CMA and NOMAC (11 cases in total) and found regression in 18% of cases, stability in 64% of cases and continued growth in 18% of cases.
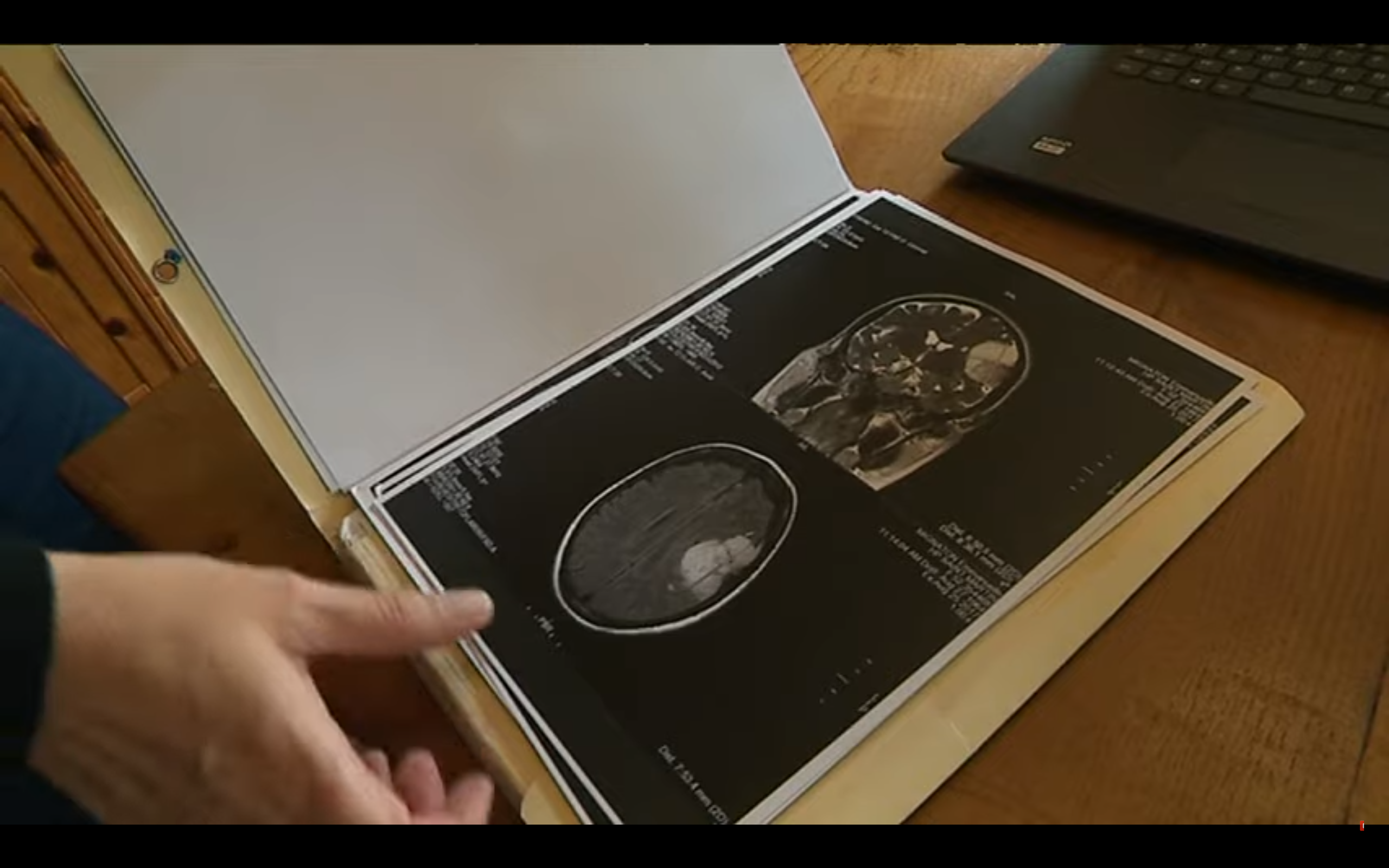
Abou-Al-Shaar et al. followed 10 women after discontinuation of MPA: in five of them, the tumors regressed; in three others, DMPA had been discontinued less than a year prior, which prevented definitive conclusions; two were lost to follow-up. Graillon et al. and Abi Jaoude et al. each reported a single case of meningioma regression after promegestone discontinuation. An illustration is presented in the supplementary data.
The evolution of osteomeningiomas after progestin discontinuation remains uncertain. Reports suggest that the dural component of the meningiomas regressed while the bony component continued to grow and even exhibited an increased growth rate after progestin withdrawal.
Location of the tumor
The published series reported a predominant location of meningiomas at the anterior and middle base of the skull, regardless of the progestin used , as summarized in Figure 4. This contrasts with the predominant location of meningiomas at the cranial convexity in the general population.
In the seven largest epidemiological studies, regardless of the specific progestin examined, the anterior skull base ranged from 20% to 37% and the mid-cranial base from 20% to 39%. In contrast, although the cranial convexity is the most frequent site in the general population, none of these studies reported a rate of convexity localization higher than that of the anterior or mid-cranial base.
Fig. 4 Tumor location. Tumor location, regardless of the progestin, is classified as convexity (A.), mid-cranial base (B.) and anterior cranial base (C.), represented respectively in blue, green and purple.