Note from the association: These measures have been taken in the “Méningiomes et Progestifififies” working group at ANSM, in which the association has sits since its creation in January 2019.
Inform that the operation is not always compulsory, because meningiomas can regress at the cessation of hormonal treatment, is also a trvail of the association since its creation.
Context
In 2018, an increased risk, dependent dose-effect, intracranial meningiomas linked to the prolonged use of cyproterone acetate in high doses (> = 25 mg/day), was quantified and made public in France. Risk reduction measures have been taken by the health authorities with information from health professionals and patients, a recall of the indications aimed at limiting the use and screening of meningiomas by brain MRI.
Objective
The objective of this study was, from data from the National Health Data System (SNDS), to measure the impact of the actions implemented to reduce the risk of meningiomas linked to the use of cyproterone acetate.
Results
Between August 2018 and December 2021 The number of people treated by high-dose cyproterone acetate decreased by 85%, with a more marked drop for women (-88%) than for men (-69%) and transgender women (-50%). In December 2021 7,900 people used Cyproterone acetate in high doses against 55,000 in August 2018 and 85,000 in January 2010. This drop resulted from a large number of treatment stops during the first wave of actions at the end of August 2018 (initial information with very important media coverage, renowning of good use and establishment of surveillance by MRI with doctors) but also during the second wave in June and July 2019 with nominative letters to doctors and patients, and the implementation of an "annual information certificate" to be co -signed by the doctor and patient. Thus almost 92% of people treated in June 2018 no longer continued their treatment in December 2021 with a marked peak after each of the waves of shares in October 2018 and July 2019. We did not find a massive postponement on a given product but a dispersion to other usual hormones (for women 35%) and the Spironolactone, anti -diarogenic effect (for women 5% and for men 3%). Cyproterone acetate establishments fell between June 2018 and December 2021 by more than 94 %.
The use of MRI brain of meningioma screening has become frequent, albeit non -systematic, with almost 70% of women user of cyproterone acetate in December 2021 which carried out this imagery and at a lower level almost 50% of men (in August 2018, before the shares these rates were 11% and 13%, respectively). For initiations of cyproterone acetate, the use of MRIs was lower, 45% for women and 32% in men.
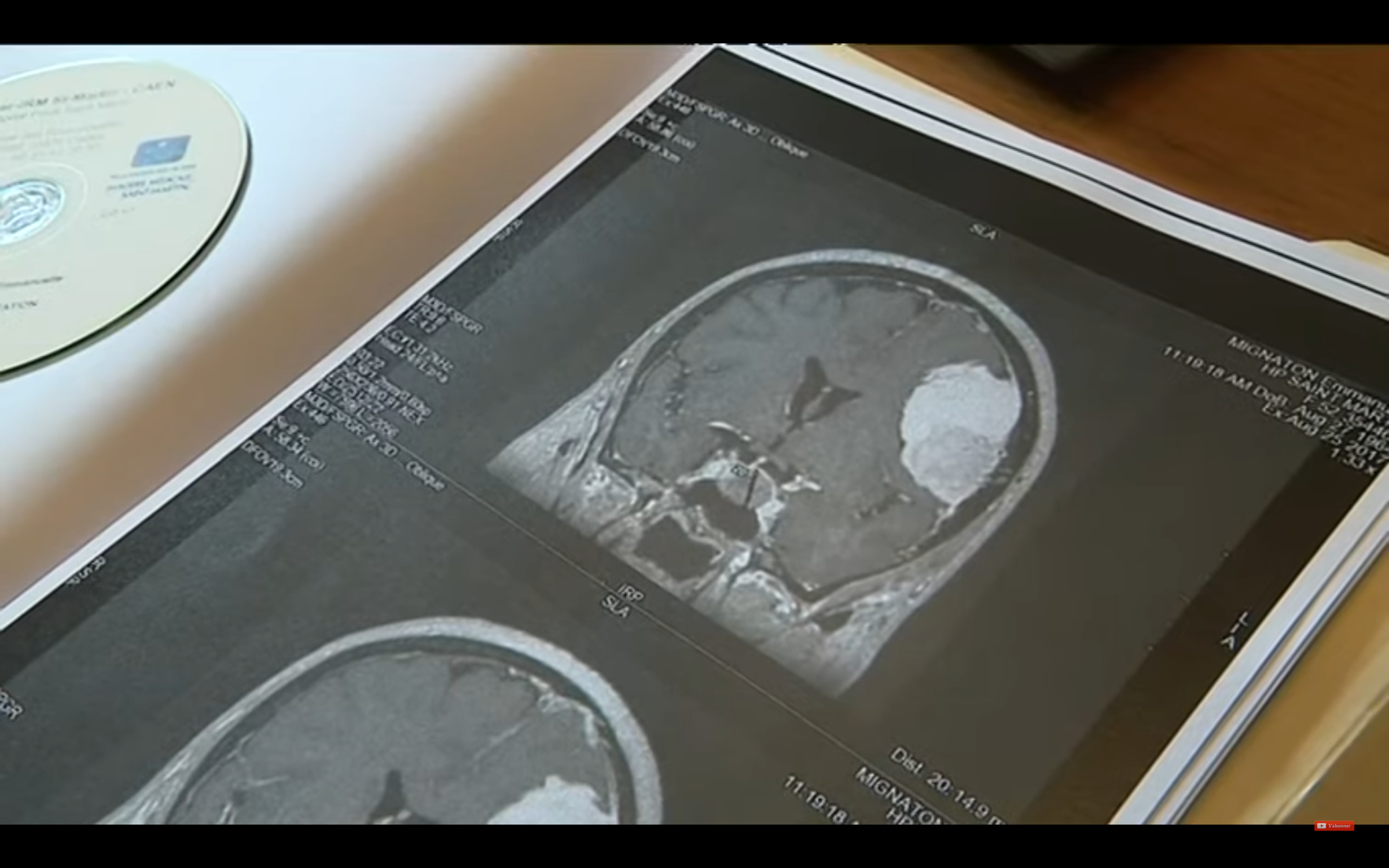
The annual number of women operated with intracranial meningioma, during or in the year following an exposure to cyproterone acetate, was thus reduced by 95 women operated in 2017 to 7 in 2021, or -93%.
Conclusion
In total, the actions carried out have greatly reduced the consumption of cyproterone acetate, which has since resulted in a collapse of the number of new intracranial meningiomas operated and attributable to this medication.