Following the announcements of the ANSM that you will find here:
https://www.ansm.sante.fr/dossiers/lutenyl-luteran/recommandations-d-lutenyl-luteran-et-leux-generiques-et-de-suivi-des-patientes/(oft)/1
We recall that the president of the AMAVEA association is part of the Experts Committee of the Working Group at the ANSM on these 2 drugs.
His latest report is here: https://amavea.org/ansm-reunion-du-16-decembre-2020-cst-lutenyl-luteran-et-risque-de-meningiome/

Article of January 12, 2021 Alert on certain progestins here: https://amavea.org/le-monde-12-janvier-2021-lutenyl-et-luteran-madicaments-pour-les-troubles-gynecologique-sont-a-utilisal-avec-precaution-en-raison-risque-de-risqueure

Top health: https://www.topsante.com/medecine/gyneco/endometriose/temoineage-luteran-lutenyl-endometriose-641185
[Testimony] "Luteran is a comfort of today and a risk for tomorrow"
Achievement of endometriosis, Axelle Ayad N'Ciri was treated at Luteran for five years. A medication that "saved his life", while exposing it to a risk of meningioma. She is a member of the scientific committee responsible for reassessing the indications for this treatment.
Axelle Ayad N’Ciri carries the voice of women suffering from endometriosis on behalf of the Endomind , as part of the public consultation on Lotényl and Lutéran treatments and the risk of meningioma. Lonely patient book and founder of Mapatho , whose objective is to relate chronic patients with the right medical interlocutors quickly, she tells Top Health her own wandering and her quest for information.
“I had my period very young when I was not 10 years old. Very quickly, I started to suffer, to have a lot of acne, to be blocked in me by pain on days of menstruation . At 14, to relieve myself, I was put under pill, without putting words on my evil. Between 14 and 23 years old, everything was fine. At least, I no longer felt the consequences of what I will learn later to be endometriosis. Until I decide to change contraception and wear a vaginal ring.
"Consult a gynecologist, there is something wrong, miss"
At that time, I am 23 years old, and my balance collapses. The pains resume more beautiful to the point that I make discomfort in my workplace. Emerming to the hospital, a doctor announces that I have an appendicitis crisis. But I consult a second, who refutes this theory: " It is not a gastro problem, it's gynecologist, you should go to consult, there is something wrong . ""
I'm going to make a radio and the manipulator said, " You may have endometriosis ". I do not know what it is, I have never heard of it, and I don't know who to turn to. So I question my entourage for three months, and we end up advising me to go to Cochin hospital, whose service specializes in infertility. My doctor, a specialist in endometriosis, offered me an immediate operation, a coeloscopy, aimed at removing the adhesions that wedge my ovaries. I am 23 years old, I was diagnosed with a disease which I do not know much and I was told about infertility . But my doctor gives me a medicine to take continuously , and tells me that I can live my life. So I go to follow my project and settle in China, as planned.
Against endometriosis, two years of luteran
This medication is luteran. Hormonal treatment that literally saves my life. I constantly take it between 2013 and 2015. I no longer have my period, I no longer have stomach pain, I have no more side effects. I'm apparently well. But this balance does not last, because after two years, my pains come back and forced me to come back to be treated in France. Adhesions (or lesions) are back, at least as strong as before.
There, the speech of doctors changes radically: it is not necessary to operate, it would risk playing on my ability to one day have children. Besides, the children, it would be necessary to think about it very quickly, I am told. At 26, I go on PMA course . But I have suffered from something else, for some time I have migraines with aura. I am discovered in addition to other pathologies: polycystic ovaries and hypothyroidism. Each PMA stimulates my ovaries and my hormones enormously and I suffer a lot. So, between two attempts (which never work), I am allowed to take over the medication that makes me my life, Luteran. I dream that the PMAs are finished to be able to be under this treatment again and stop suffering.
The impression that my brain is too big for my skull
In 2020, I definitively give up the PMAs and I ask the doctor to get back under continuous Luteran. My migraines come back hard, I have the feeling that my brain is too big for my skull. My doctor tells me that things are starting to emerge on this medication, which we question ourselves about its side effects ... But at that time, I say to myself: I have no other way to relieve endometriosis and the Nurofen helps me to resist migraines. So I continue. I try other treatments, but the side effects are terrible: weight gain, disabling pain, continuous rules, acne ... Nothing works as well as luteran. After the wandering of diagnosis, wandering against infertility, I saw drug wandering.
That said, I learn more about this medication, which can cause meningioma (brain tumor) . Benin in the purely medical sense of the term but certainly not without gravity or consequences since it can cause pain, epilepsy, vision disorders ... I must choose between plague and cholera: everything stop and let endometriosis progress, take luteran and risk a tumor in the brain, or choose another treatment whose effects are unbearable.
Tomorrow, what solutions for patients?
I represent Endomind within the public consultation on the risks linked to Lotéran and Lentenyl (Editor's note, the ANSM issued new recommendations this January 12). Luteran is comfort today but a huge risk in the future. But what are the alternatives for women who suffer from endometriosis? My goal is to bring the voice of women who take this drug so that they have access to all the necessary information and that they are entitled to a tailor -made medical journey, with regular MRIs to monitor the possible birth of a meningioma. »»
Our time
https://www.notretemps.com/sante/risque-de-meningiome-usage-restreint-fp-202101,i235213
The use of progestinic drugs, which present an over-risk to develop a cerebral tumor -Méningioma-must now be restricted to certain indications, be as short as possible and if not, its merits must be reassessed "at least every year", according to the ANSM drug agency.
These treatments were taken by more than 400,000 women in 2019 for menstrual disorders or menopause and endometriosis.
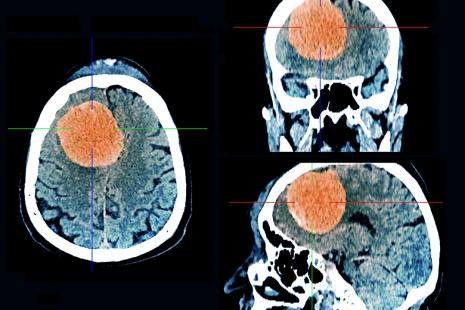
Meningioma is a tumor of the brain envelopes, the vast majority benign.
Updating the recommendations published Tuesday by the ANSM concerns Lotéran (Chlormadinone acetate) and Lotényl (nomestrol acetate) and their generics as well as the follow -up of the women concerned.
These recommendations, which replace those, provisional, of June 2020, relate in particular to the situations in which the benefit-risk report of these treatments is favorable as well as on the monitoring of patients by brain imaging (MRI exam).
When the alternatives have failed or are contraindicated, these drugs can be prescribed in certain cases such as bleeding linked to pre-operative fibroids or other origins ("functional hemorrhages"), or even anomalies in the breasts ("severe mastopathy").
Luteran generics can also be considered for endometriosis. The treatment must be as short as possible and in the opposite case the benefit-risk report reassessed at least every year.
On the other hand, these treatments, compared to the risk of meningioma, should no longer be prescribed in particular as contraception - "many women took it as a conventional pill" -, or for menopause disorders, notes with AFP Dr Isabelle Yoldjian, head of the ASM gynecological pole. Especially since therapeutic alternatives exist and the risk already increases with age, regardless of any treatment.
Likewise, "the irregularities of the cycle, that is to say when a woman who has cycles not quite regular, or when she has a stomach ache during her period, are indications that we should no longer legitimize," she adds.
With these recommendations, the downward trend in the sale of these drugs should increase, she notes.
According to the update, monitoring by brain imaging examination (MRI) should be carried out regardless of the age of the patient at any time during or after treatment if she has signs evocative of meningiomas (frequent headaches, vision disorders, language, dizziness, etc.) and no longer just from 35 years old. If the treatment is continued, MRIs are planned.
These drugs are already contraindicated in the event of existence or history of meningioma. The treatment must be stopped immediately and definitively in the event of diagnosis of this tumor.
A meeting of the Committee of Experts will be held on January 22 to discuss information documents intended to support these recommendations.
The risk of meningioma, confirmed by a vast epidemiological study published in June 2020, increases with large doses and long -term treatment. A warning on this possible risk had occurred in February 2019 several months after drastic supervision measures for the same reasons towards the Androcur and its generics (active principle: Cyproterone acetate).
The daily newspaper
Genethics
https://www.genethe.org/lutenyl-luteran-et-risque-de-meningiome-lansm-revise-ses-Recommandations/
The National Medicines Safety Agency (ANSM) has updated its recommendations concerning the use of "progestin drugs" . Despite their "over-risk of developing a brain tumor" confirmed in June [1] , the health authority "did not deem it necessary to withdraw the products" . It restricts their use "to certain indications" [2] , for a "shortest possible" or at least a reassessment of the "well-founded" of its administration every year. Their prescription for contraception, menopause, irregular cycles or pain of non -severe breasts is prohibited. The brain imaging of women taking these progestins over more than a year is no longer linked to age.
The two targeted specialties are Luteran (Chlormadinone acetate) and lutenyl (Nomestrol acetate), as well as their generics, whose use is "massive in France": "In 2019, around 200,000 women took nomestrol and 240,000 from Chlormadinone acetate, which represents 80 % of the prescriptions of these molecules in Europe". The Androcur (Cyproterone Acetate) had also been the subject of studies in 2019 and a restriction of its indications (cf. contraception and brain tumors: the drug will not be removed from the market ; Androcur, used as contraceptive, causes tumors to the brain but continues to be authorized , Androcur: new cases of tumors, a legal action ). For Professor Froelich, neurosurgeon in Lariboisière (Paris), "all progestins are involved in meningiomas, to various degrees". At the Brest CHRU, in addition to Androcur, Lotényl and Luteran, the IUD Mirena, the Duphaston, Diane 35 and the Progestogel have been the subject of pharmacovigilance declarations linked to the Meningioma and Progestifs Association. "A study on Mirena has just started" .
The Committee of Experts must meet on January 22 "to discuss information documents intended to support these recommendations" . In the 315 testimonies received by the ANSM and made public, the "lack of information" of women "returns repeatedly".
[1] The study published in June 2020 [1] shows that "the risk of meningioma for women who have taken these products between six months and ten years is tripled compared to unpresentation women" . A risk that "increases sharply with the cumulative dose, the duration and age of the patient" (cf. over-risk of brain tumors linked to the taking of progestin treatment ). Note that the link between progestins and meningiomas "is not new", already highlighted by previous studies.
[2] "When the alternatives have failed or are contraindicated, these drugs can be prescribed in certain cases such as bleeding linked to pre-operative fibroids or other origins (" functional hemorrhages "), or even anomalies in the breasts (" severe mastopathy "). Luteran generics can also be considered for endometriosis. »»
Sources: AFP (12/01/2021); Le Monde, Pascale Santi (12/01/2021)
Yahoo News : HERE

Current woman
In June 2020, a study by the National Medicines Safety Agency (ANSM) revealed the risks of brain tumors associated with the use of luteran and lutenyl, namely progestin drugs. The health authority unveiled new recommendations on these treatments on January 12.
Luteran and Lotényl are two progestin drugs, derived from progesterone. They are indicated in the management of menopause , uterine fibroids , endometriosis and monthly disorders, such as painful rules or cycle irregularities.
These drugs, which have been administered as a contraceptive pill or as a treatment of acne , would increase the risk of meningioma in women, according to the National Agency for the Safety of Medicines (ANSM). Meningioma is a brain tumor , which can be responsible for serious disorders and may require heavy and risk surgery.
In a vast epidemiological study, published in June 2020, the health authority revealed that women who take one of these progestins for more than six months would have approximately 3.3 times more risk of developing meningiom compared to the basic risk. The risk is multiplied by 12.5 for those who take lutenyl for five years. When Luteran is used for three and a half years, it multiplies the risk of meningioma by 7.
Lutéran and Lotényl: What are the new ANSM recommendations?
Given the “demonstrated sur-risk of meningioma associated with the taking” of Lotényl and Lutéran, the ANSM judged that these drugs can no longer be prescribed under the same conditions and updated its recommendations , which were published this January 12. The authority has decided to restrict certain indications for these progestins and to define the conditions for setting up monitoring by MRI .
cycle irregularities , premenstrual syndrome contraception mastodynies because for these situations, "benefit/risk is considered unfavorable ," said authority.
On the other hand, these drugs can be administered in case of functional hemorrhages, menorrhagia linked to fibroids , severe mastopathy, endometriosis because the benefit/risk is "favorable". But this will only be possible for "women of childbearing age when the therapeutic alternatives have failed or are contraindicated" . The ANSM has specified that, in this case, the treatment must be as short as possible and that the benefit/risk ratio must be reassessed at least once a year.
The agency also recommends that an MRI is carried out by all women who take these two progestins, regardless of their age. This examination should be carried out during or after treatment in the event of symptoms evocative of meningiomas. The examination is recommended after a year of treatment when it requires being prosecuted, then 5 years after the first MRI, then every 2 years as long as the treatment is continued. MRI should also be carried out "in the initiation of treatment in the event of identified risk factors of meningioma", detailed the authority.
What to do if we take or if we were treated with lutenyl and luteran?
The ANSM invited women, who take or took these progestins, to be attentive to the evocative signs of meningioma, namely frequent headaches vision , language or memory , and to discuss their care with their doctor. "You can be brought together to develop your treatment and/or set up a follow-up by brain imaging according to your situation," read the agency's press release.
- In June 2020, an epidemiological study revealed an overview of developing meningioma for women treated in Lotéran and Lotényl, two progestin pills used by around 600,000 women in France.
- In September, the ANSM launched a public consultation and organized public hearings of patients to see in which cases these treatments were prescribed.
- 20 minutes retraces these few months of health democracy by giving the floor to the head of the ANSM gynecology pole and one of the patients interviewed.
The survey will have taken six months, but the recommendations may strongly change their use. After having alerted in June 2020 to the risk of meningioma (most often benign tumor of the brain) for women who take luteran (whose active molecule is chlormadinone acetate) and lutenyl (nomigestrol acetate), two very common progestive drugs, the national drug safety agency (ANSM) unveils its new recommendations on Tuesday.
Meanwhile, the agency has requested caregivers, patient associations, researchers and women concerned to stick as close as possible to the field.
What indications and what follow -up?
Result: ANSM clearly limits the indications. These pills should no longer be prescribed in the event of menopause, irregularities in the menstrual cycle, premenstrual syndrome, light mastodynia (breast pain) and contraception. On the other hand, the benefit/risk balance was deemed favorable in the event of endometriosis , functional hemorrhages, menorrhagia (very abundant rules) linked to fibroids and severe mastopathy (non -cancerous breast disease).
"For Lotéran and Lotényl, marketing authorizations (AMM) date from 1973 and 1984 respectively," says Isabelle Yoldjian, head of the ANSM gynecology pole . However, they are massively used with, in a good number of cases, a relative deviance compared to the Label of the AMMs. It was important to clean up these indications. »»
How many women would be affected? The work of the ANSM did not make it possible to obtain exhaustive figures, but "we know that in 2019, around 200,000 women had taken lutenyl (or generics) at least once and 240,000 of the luteran (or generic). Suffice to say that these new recommendations, if they are followed, may drop sales. "Between August 2018 and August 2020, there is a decrease in prescriptions of 30 % for the Lotényl and 50 % for the Lotéran," says Isabelle Yoldjian.
Second issue: what will be follow -up for patients? Again, the recommendations are clearly moving away from what had been going on so far. “In June, we recommended to make magnetic resonance imaging (MRI) from 35 years old . There, we removed the age barrier, she underlines. All women must do brain MRI from one year of treatment. Then the patient is encouraged to make another MRI five years after the first imagery. Then every two years.
Why this acceleration? "If you take treatment for three or four years, the risk is less than eight years," says Isabelle Yoldjian. The longer you are treated, the more close surveillance it takes. And of course, all patients must carry out this examination at any time during or after treatment in the event of signs evocative of meningiomas (headache, language disorder, vision, memory, paralysis, dizziness).
"It was presented to me as the miracle solution"
To avoid missing out on an important subject, patients have been able to give their opinion on these treatments in recent months. In particular via a call for testimonies (which received 500 contributions) and six public hearings of patients with different profiles. Véronique, 30, is one of the patients consulted. After receiving the diagnosis of endometriosis at 28 years old, and suffered martyrdom, she took luteran for nine months, then lutenyl for a year. "It was presented to me as the miracle solution," she explains. And it's true that it helped me on pain. But the side effects were very important: acne, weight gain, depressive symptoms ... "In June 2020, following the ANSM alert, his surgeon advised him to stop immediately. "It is a shame to say that doctors, researchers were aware of the risks and that I learned it by the press," she regrets. So when the Endofrance warns that the ANSM is looking for testimonies , it proposes.
"I am not saying that all women experience Luteran badly, but it was my case. I was so sensitive that I could cry if someone did not pass me salt at the table! When I complained of having taken 12 kg, I was told that I may have ate too much? On November 2, during public hearings (in Visio, Cavid-19 obliges), Véronique was unable to listen to all the testimonies, "so they (l) 'destabilized. Some stories are dramatic. One of the patients had undergone invasive brain surgeries. All that to better live menopause… ”
Not "a banal pill"
The speech of the patients has reserved some surprises for the experts of the committee. Especially on the use of these treatments as contraception. " Following the release of the first article in 20 minutes , many women responded to our call for testimonies and thought that their treatment was a commonplace, is surprised Isabelle Yoldjian. Public auditions have been particularly interesting on this aspect. »»
Hence the watchword that the agency wishes to pass: "In contraception, there is no need to authorize this treatment insofar as there are alternatives," she insists. Likewise for menopause. “The risk of meningioma increases with age. We therefore come up against a very important impact if we combine this risk with the use of a drug that gives meningiomas. The second thing that has come back from hearings is the lack of information available to women. Health professionals are not aware or do not transmit information, which is recent, it is true. These testimonies encouraged us to email healthcare professionals in November ”.
How to improve information to patients?
The agency intends to go further. A final meeting of the Committee is scheduled for January 22 to find out how caregivers and women can have the right information. For the Androcur ( another progestogen which had been the subject of a public consultation ), the agency had set up a certificate: during the prescription, caregiver and patient had to sign a document specifying that the latter was aware of the risk incurred. Which certificate is compulsory to buy Androcur. Will this be the case? The question remains to be decided. "What we know is that an information letter will be sent to all the patients who receive Lotéran and Lotényl," reveals Isabelle Yoldjian.
Véronique hopes that this public consultation will allow "to inform patients much better. Most of the testimonies I heard mentioned this lack of alert. We often advance the question of profit/risk, but we can only hear about it in a gynecologist! And beyond these progestogens, it would be necessary to pay attention to the patient's information when it is given a new treatment. »»

Risk of brain tumor: Lotényl and Lutéran in the viewfinder of the Medicines Agency
The medication gendarme has issued new recommendations on the use of Lotéran and Lotényl, two drugs that are very common.

By Maxime François on January 18, 2021 at 8:48 a.m., modified on January 19, 2021 at 06:27
Alert on drugs (Nomestrol acetate), Lutéran (Chlormadinone Acetate) as well as their generics - progesterone derivatives - intended for gynecological disorders! They are to be used with great precautions in the face of the increased risk of brain tumors that they can cause, alerts the drug agency (ANSM) after having carried out a large investigation into these treatments intended to treat menstrual disorders, menopause, or premenopause, endometriosis, but also, and less, as contraceptives. The subject is hot: in 2019 alone, nearly 400,000 women consumed these drugs.
From now on, their use must be restricted to specific indications and be as short as possible. Otherwise, their consumption must be reassessed "at least every year" because "the risk increases sharply with the cumulative dose, the duration and age of the patient", specifies the study. It is even multiplied by 12.5 from five years of treatment under Lotényl, and by 7 after three and a half years under Lutéran.
This study focused on 1.8 million women who have taken nomigestrol acetate and 1.5 million chlormaudinone acetate, between January 1, 2007 and December 31, 2018. In total, "more than 1000 meningiomas ( editor's note: tumors of the brain envelopes, the vast majority benign ) operated are attributable to these two molecules between 2007 and 2018 Case per year, "revealed epidemiologist Alain Weill, deputy director of Epi-Phare, who piloted the study.
Follow -up by MRI
Faced with these new results, the ANSM has issued new recommendations, including monitoring patients by brain imaging (MRI) . These examinations should be carried out regardless of the age of the patient "at any time during or after treatment in the event of signs evocative of meningiomas ", "after a year of treatment when the latter requires being prosecuted, then five years after the first MRI, then every two years as long as the treatment is continued" but also in the event of "identified risk factors", warns the ANSM.
The latter specifies that for women in a situation of "menopause, artificial cycle in association with an estrogen, cycle irregularities, premenstrual syndrome, non-severe mastodynia or contraception (without factor of associated cardiovascular risk)", "benefit-risk is considered unfavorable" and we must not use these treatments.
When the therapeutic alternatives have "failed" or are "contraindicated", the "benefit-risk is considered favorable" for the use of lutenyl for women knowing "functional hemorrhages, menorrhagia linked to preoperative fibroids or severe mastopathy," said the agency. For the Luteran, the criteria are similar but the ASNM specifies that the use of this treatment is also "favorable" for women with endometriosis . Newsletter most in the morning. A turn of the news to start the day .
These recommendations follow a previous study by ANSM and published in June . It already established the sharp increase in the risk of meningioma in women taking these treatments. An expert committee must meet on January 22 to discuss information documents intended to support these recommendations.