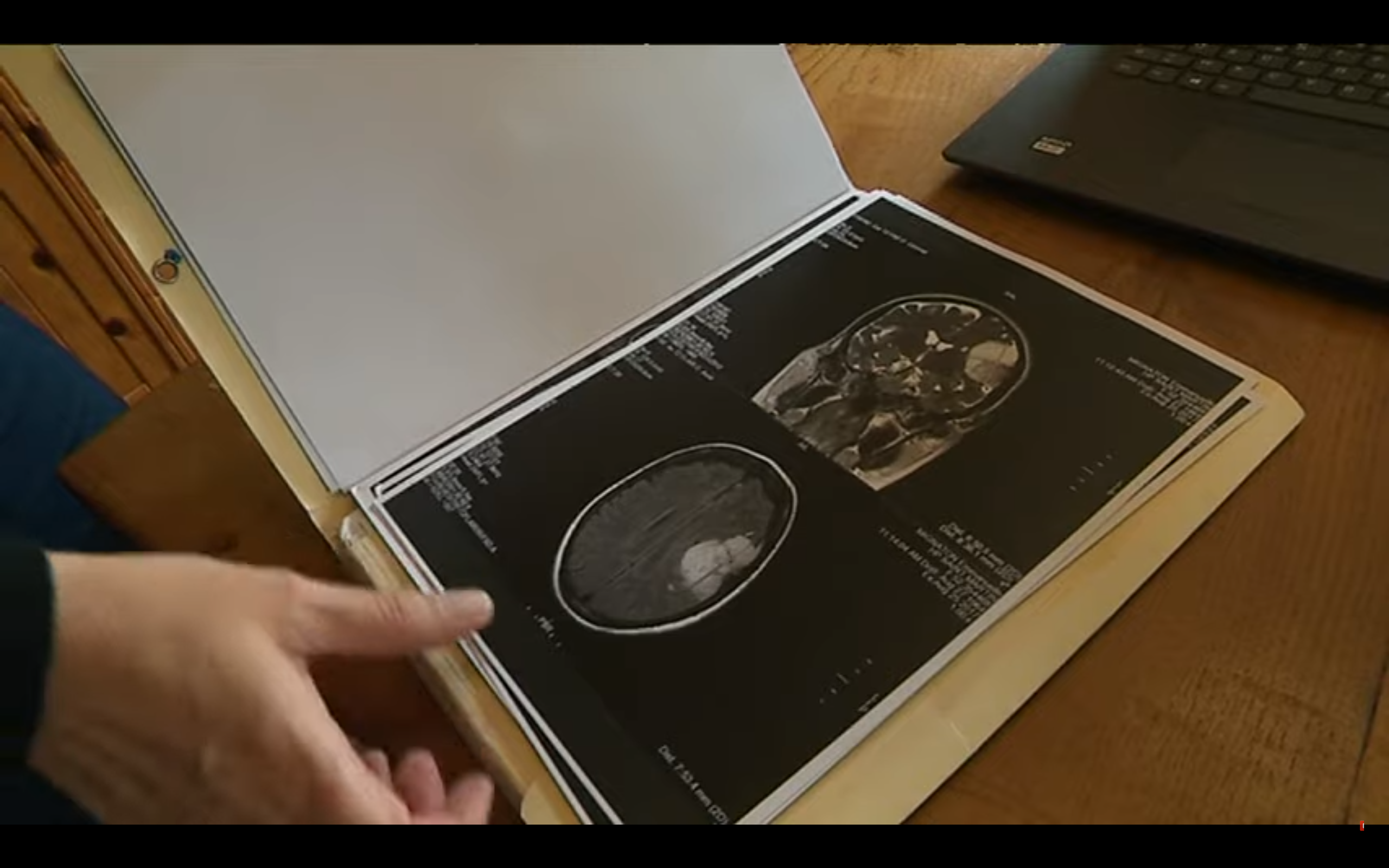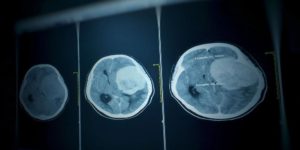
However, all the women affected by these treatments indicated in the management of menopause, menstrual disorders and endometriosis, are not yet informed of this risk. This is why the National Medicines Safety Agency (ANSM) has decided to deploy a new information system to patients and health professionals.
Information and certificate
First, the agency will provide caregivers with an information document to be given to each patient to start processing or pursue it. Its content? Explanations on what a meningioma and the level of risk is depending on the treatment taken. "He will alert clinical signs likely to be associated with the presence of meningioma and will specify the driving to be held for each situation," notes the ANSM.
In parallel, each patient and her doctor will now have to complete an annual information certificate. The goal is to ensure that the patient is well informed by her practitioner of the risks involved. This certificate must be presented in pharmacies before obtaining treatment.
These documents, inspired by those implemented in 2019 to reduce the use of another progestin medication associated with an over-risk of meningioma (Androcur and its generics), will be available during spring. If certain methods remain to be defined, the objective of these measures is the same here: "Limiting the use of these drugs to the only situations for which their prescription is justified", specifies the ANSM.
In the meantime, health professionals have already been encouraged since June 2020 to:
-reassess the relevance of one of these treatments taking into account the individual benefit-risk;
- Limit the duration of use of these drugs as well as their dosage at minimum effective doses;
– Do not substitute macroprogestatives between them (nomestrol acetate, chlormadinone acetate and cyproterone acetate) in the event of meningioma or a history of meningioma ;
- have a brain MRI made in the event of symptoms evocative of a meningioma;
- Offer women over 35 in the event of prolonged treatment (from 5 years old), brain imaging by MRI.
To find out more, do not hesitate to consult the questions/answers of the ANSM: https://www.ansm.sante.fr/dossiers/lutenyl-luteran/questions-sur-les-risque-de-meningiome-sous-lutenyl-luteran-leux-generiques/(oft)/2
*Study carried out from national data insurance reimbursement data on 1.8 and 1.5 million women who have consumed nomestrol acetate or chlormadinone acetate between January 1, 2007 and December 31, 2018
Link to the article here: https://www.sudouest.fr/sante/luteran-lutenyl-la--frevention-du-risque-de-meningiome-renforcee-1367114.php
Find our other articles here