Epilepsy linked to meningioma - Physiopathology, predictors and treatment of pre/postoperative crises
by Rasha Elbadry Ahmed1 †, Hailiang Tang2 †, Anthony Asemota1, lei Huang1,3, Warren Boling1*‡ and Firas bannout4*‡
-1 Neurosurgery, Loma Linda University Medical Center, Loma Linda, CA, United States
- 2 Neurosurgery, Huasha Hospital, Fudan University, Shanghai, China
-3 Between physiology and pharmacology, University of Loma Linda, Loma Linda, CA, United States
-4 Neurology departure, medical center of the University of Loma Linda, Loma Linda, CA, United States
Meningiomas are the most common primary brain tumors and represent approximately 30 % of all brain tumors . The vast majority of meningiomas have slow growth and benign histopathology, which makes them curable by surgery alone.
Symptomatic lesions depend on their location with signs of mass effect or neurological deficits.
Epilepsy attacks are the main symptom in around 30 % of cases, which negatively affects quality of life, limits independence, alters cognitive functioning and increases the risk of psychiatric comorbidities, including depression.
Although surgical resection can provide an absence of crises in 60 to 90 % of meningiomas, crises persist after surgical resection in approximately 12 to 19 % of patients .
Anti-convulsion drugs (ASM) are used in management, but they are limited by unwanted neurocognitive side effects and their ineffectiveness in certain patients.
Potential predictors of pre -and postoperative crises in patients with meningioma have been identified in the literature.
Understanding the various factors associated with the probability of crises in patients with meningiomal can help guide more effective control of crises and allow better risk determination before and after surgery.
Introduction
Meningiomas represent approximately 30 % of primary brain tumors and approximately 54 % of primary benign tumors (1-3). The vast majority of meningiomas are of slow evolution and benign histopathology (that is to say tumors of grade I of the World Health Organization (WHO)), which makes them curable by surgery alone (4, 5). Symptomatic lesions depend on their location with signs of mass effect or neurological deficits. Epilepsy attacks are the initial symptom in around 30 % of cases, and in certain studies, the percentage varies from 13 to 60 % (6-8). Although surgical resection can provide an absence of crises in 60-90 % of meningiomas, crises can persist after surgical resection in approximately 12-19 % of patients (9, 10). The crises can have a negative impact on the quality of life, by hampering the patient's independence, his cognitive functions and his ability to drive in complete safety (11-13). They expose patients to an increased risk of different psychiatric comorbidities, including depression (14). Crisis control using various anti-crisis drugs (ASM) is generally offered despite the undesirable side effects on neurocognition and ineffectiveness in certain patients (15).
Many theories have been put forward to explain the pathogenesis of epilepsy linked to brain tumors (BTR) in various brain tumors; However, questions remain unanswered about the control and management of crisis in patients with meningiomas, for example, the ability of surgical resections to cure crises, when starting ASMs, the duration of treatment as well as structured directives for the selection of patients for ASMs. Understanding and predicting crises in meningiomas can help guide crises control and better determine patients at risk before and after surgery. The purpose of this review aims to summarize the pathogenesis of crises in meningiomas, predictive predictors and postoperative crises, surgical resection which eliminates crises, the advantages of the use of ASMs, peroperative electrocorticography (ECOG) and monitoring of electroencephalogram (EEG) in patients with patients with patients with patients meningiomas and the appropriate selection of patients.
Incidence of epilepsy in meningiomas
The incidence of preoperative crises in the case of meningiomas has been reported respectively at 29 % out of 4,709 patients (7) and 14 % out of 598 patients (16) with supratotory meningiomas.
The absence of crises was obtained in around 69 % of patients after surgery , with 12 % of new crises occurring after surgery (17). Chozick's study reported that 63/158 patients with meningioma had preoperative crises and that 40 (63.5 %) of the 63 patients had complete crises after surgery in the years of monitoring of 7.3 ± 3.8. In this cohort, 100 % of the 63 patients were under anticonvulsant treatment before the operation and during the initial postoperative phase. The authors did not indicate the exact proportion of these 43 patients wealed from anticonvulsant drugs over time. While some neurosurgeons tended to stop using the drugs about 6 months after the operation if there were no sign of crises, the other neurosurgeons continued to use anticonvulsants on a prophylactic basis. They reported that eighty-five patients (53.8 % of 158) were finally wealed from anticonvulsants and that 44.7 % of them had not stopped taking them during the last postoperative follow-up visit. The crises are reappeared in a patient during the ASM withdrawal process, in 4 patients with sub-therapeutic ASM levels, in 6 patients who were not on ASM, in 2 patients correlated with alcohol abuse and in 5 patients with tumor recurrence. Eight patients (5.1%) without history of preoperative epilepsy developed postoperative crises. Chozick et al. concluded that in their study, only the extent of the tumor's ablation was a significant predictor of postoperative crises .
However, history of preoperative epileptic crisis, preoperative language disorders, postoperative status of anti-crisis drugs, postoperative hydrocephalus or the location of tumor in the parietal region were also predictive factors for the occurrence of postoperative epileptic crises (18). Wirsching reported 26.6 % postoperative crises during a median period of 67 months (95 % CI: 63-72) postoperative monitoring (19).
The great variation of these studies can be due to the lack of standardization of the data collected retrospectively in patients with different demographic characteristics, characteristics/locations/types of different meningiomas, different monitoring periods, analyzes of different age groups between pediatric and adult patients, and different surgical skills and techniques in different institutions. The majority of postoperative crises were felt during the first week after the operation, but a third of patients presented crises three months after the operation (17).
Pathogenesis of epilepsy linked to meningioma
The pathophysiology of epilepsy linked to a brain tumor is multifactorial and can be divided into morphological, biochemical and metabolic causes.
The morphological changes in the peritumoral neocortex include the connection of neurons and the connectivity and location of synaptic vesicles, resulting in a higher concentration of NA+ voltage channels, Ca ++ and glutamate receptors with a loss of inhibitory synapses and an increase in excitatory synapses. Biochemically, there is an increase in glutamatergic neurons and a reduction in immunoreactive gabaergic neurons to somatostatin. At the ionic level, we observe a low rate of MG2+, a high rate of extracellular K+, a high rate of Fe3+, a low rate of K+/Cl- Cotransporter-2 (KCC2) specific to neurons. It is believed that the peritumoral extracellular pH is slightly alkaline. Finally, there are enzymatic changes, amino and immunological acids with upward regulation of glutamatergic receptors for neurotransmitters NMDA and AMPA (24).
More recently, the genetic factors of the epileptogenicity of meningiomas have been studied. The NF2 mutation has proven to be a predictive marker of preoperative crises, via an indirect mediation effect with atypical histology and edema (25). Meningioma comes from cells in the arachnoid cap and is generally a slow growth tumor (1). This slow growth can partly explain the peritumoral modifications which lead to epileptogenicity (24, 26). Partial differentiation of the cortical surface of the brain can produce an epileptogenic area, thus causing denervation hypersensitivity (27). The morphological changes that develop in the cerebral fabric adjacent to the lesion, such as ineffective neuronal migration, synaptic vesicles and alterations of the Gap-Junction Glial coupling, are also supposed to contribute to the generation of crises (28). Although pediatric meningiomas are rare, epilepsy has been reported as one of the current symptoms (29). A ineffective neuronal migration could constitute an additional peritumoral mechanism of epileptogenesis in this age group of patients.
The percentage of brain edema in patients with meningioma varies between 30 and 60 % (30-32). It is generally vasogenic and linked to an increase in PIAL intake, angiogenesis and the increased expression of the vascular endothelial growth factor (VEGF) (33, 34). Chemical changes in the peritumoral environment and local hypoxia due to the local compression of the tumor are considered as underlying mechanisms which decrease the threshold of crises (26). The increase in glutamate levels in peritumoral edema is often described as an instigator factor in the state of hyperexcitability and epilepsy (8, 26).
The edema is strongly correlated with the invasion of the brain (35), and can also be intimately associated with the location of the tumor and with more invasive meningiomas of higher grades (31, 32, 36). In particular, Hess et al. Reported a five -year multiplication of edema in patients in patients with cerebral invasion compared to those who do not present, with a 20 % increase in the risk of cerebral invasion for each increase of 1cm of peritumoral edema (35). Chernov, et al. reported a high incidence of peritumoral edema in macroscopic invasive meningiomas (37). The cerebral invasion and the rupture of the arachnoid layer deform and alter the peritumoral cortex, releasing amino acids and affecting the path of neurotransmitters (35, 38).
With regard to the appearance of postoperative crises, strong peroperative memberships , the need to carry out a microdissection, as well as any lesions of the cortical surface and irritation can contribute to the generation, in particular in patients who have never had crises (36). Retraction and manipulation, which are sometimes necessary to obtain total resection in the base of the skull, can also lead to additional cortical lesions and edema (39). Postoperative complications such as infection, hematoma and hydrocephalus can further increase brain edema and increase the risk of crises (40).
Based on histopathological characteristics, the WHO classification system classifies meningiomas in Grade I (Benin), Grade II (atypical) and Grade III (anaplastic) (41). Hess et al. have retrospectively analyzed the cerebral invasion and the risk of convulsions in a total of 176 patients with meningiomas. There were 92 (52 %) Grade I, 79 (45 %) tumors of Grade II, and 5 (3 %) of Grade III. Grade I meningiomas included 16 (17 %) transitional subtypes, 4 (4 %) secretory, 68 (74 %) meningothelial, 3 (3 %) fibrous and 1 (1 %) angiomatous. Preoperative crises were present in 10 (11 %) of the 92 patients with grade I, 23 (29 %) of the 79 patients with grade II meningioma, and absent in patients with anaplastic meningioma. In the meningiomas of Grade I, the histopathological subtype was significantly correlated at the rate of preoperative epilepsy. Overall, the risk of preoperative epileptic crisis was significantly higher in patients with a tumor of grade II or III than in those with a grade I tumor. Brain invasion was absent in all patients with grade I meningioma, but it was present in 35 (44 %) of patients with atypical meningioma and in 3 (60 %) Anaplastic meningioma.
The brain invasion was independent of the volume of the tumor but strongly correlated with the volume of the edema. Multivariate analyzes have shown that the risk of preoperative crises was much higher in patients with meningioma with cerebral invasion than in those with non-invasive meningioma (OR 5.26, 95 % 1.52-18.15 CI; p = 0.009).
However, rates of postoperative crises were similar in patients with invasive meningioma and those with non -invasive meningioma. The incidence of postoperative epilepsy was significantly correlated with the increase in preoperative tumor volume (35). In another retrospective study, Gadot et al. examined the 384 patients who underwent a resection of meningioma. The significant association has not been found between a histological subtype and more bad postoperative results in terms of epilepsy attacks. However, there was an associative tendency between higher grades of grades (clever, rhabdoid) with worse postoperative results in terms of crises. The subtypes of lower grades (fibrous, transitional) tended to improve postoperative results (p = 0.081) (25). There are no data in the medical literature for little fortuitous meningiomas, which are not part of the epileptogenic network.
Predictive factors of epilepsy in the case of meningioma
In order to better understand and predict epilepsy attacks in patients with meningioma, several retrospective studies have examined the possible predictors of crises both preoperatively and postoperatively. Throughout the literature, peritumoral edema and the location were associated with crises in meningiomas. Peritumoral edema has been the subject of in -depth studies and is considered to be the strongest predictor of crisis both preoperatively and postoperatively (7, 8, 17, 20, 21, 26, 35). The probability of obtaining an absence of a postoperative crisis is less in patients with significant preoperative edema (21, 42).
Predictive predictive factors
The predictive predictive factors of epilepsy/convulsions are summarized in Table 1.
Table 1
Table 1 predictive factors of epilepsy/preoperative crises.
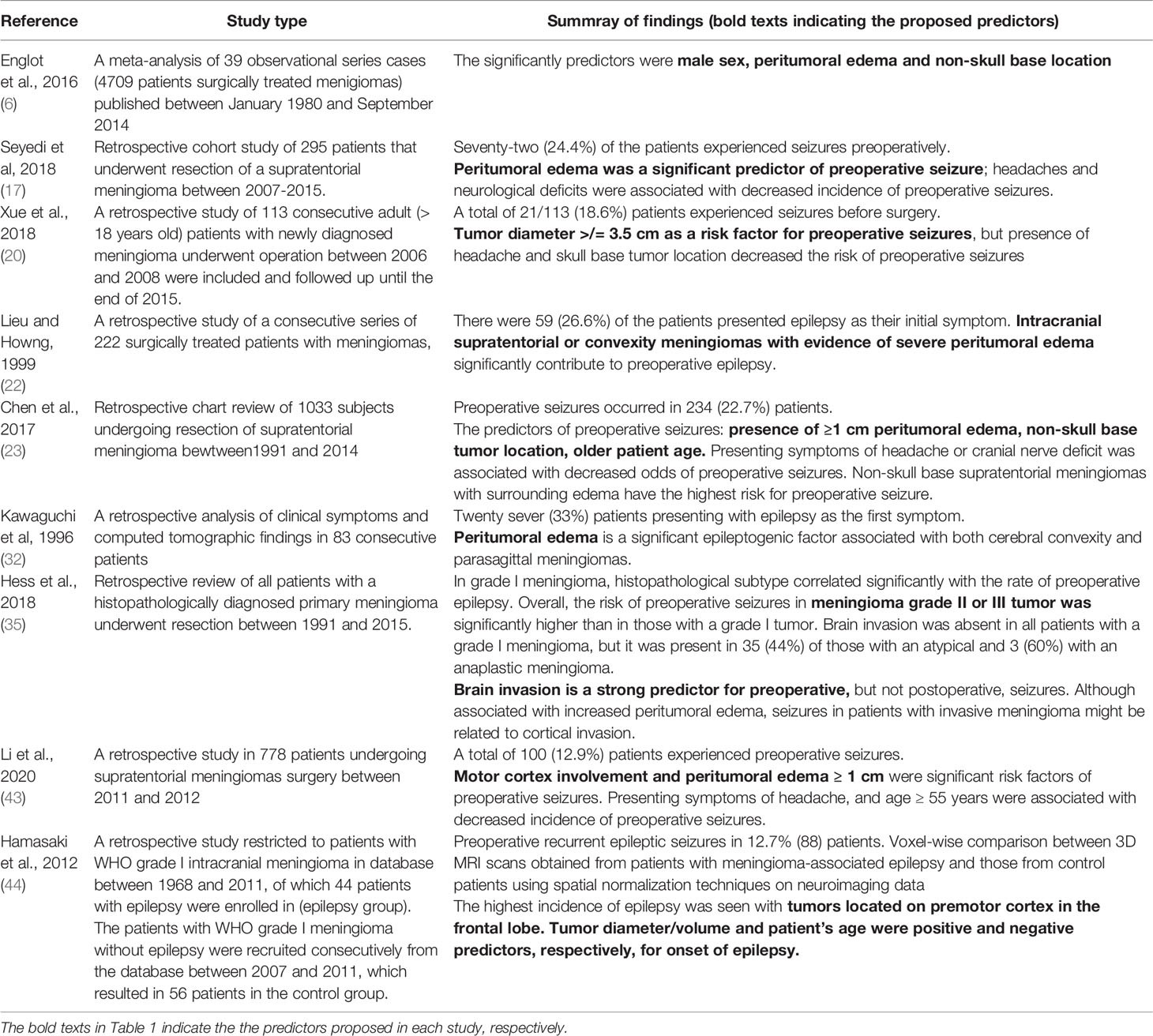
In a retrospective study of Li et al. The peritumoral edema of> 1cm was among the risk factors identified for preoperative crises in patients with meningioma (43). The location of the tumor in temporal, parietal and frontal lobes (adjacent to the neocortex) is more likely to be associated with crises (7, 18, 20, 21). More specifically, place and Howng noted that the tumor located in the temporal lobe increased the risk of preoperative crises compared to other lobes. It is believed that the increase in peritumoral edema noted in convex and parasagittal meningomes promotes the probability of an increase in the frequency of crises in affected individuals. Meningiomas that are not at the base of the skull are suggested as more aggressive with a high MIB index (percentage of immunoreactive tumor cells) which promotes brain invasion, edema and crises (20, 45). In another study, no consensus was found concerning the most epileptogenic cortical area (46).
Most studies suggest that larger tumors are naturally associated with a higher risk of preoperative crisis. It is conceivable that larger tumors can cause more irritation and compression on surrounding brain tissue. Similar results reported by Chen et al. have shown that tumors over 3 cm, of upper grade with peritumoral edema of more than 1 cm are associated with preoperative crises (20). In a study, no statistically significant correlation between the size of the tumor and the preoperative crises could be found (43), while the average diameter of the 3.5 cm tumor was used as a threshold to demonstrate an association with postoperative seizures in the hospital.
It is interesting to note that meningiomas are more frequent in women, but that men are more likely to present epilepsy attacks. Many studies have shown that male sex is a risk factor in developing preoperative crises (7, 8, 20, 23, 43). There is a possible association between male sex and higher grade meningioma, larger size and greater edema (20). A younger age was a predictive factor (44), and a lower impact of preoperative crises was found in patients with meningiomas aged over 55 (43).
Other factors such as preoperative Karnofsky (KPS) have also been studied. A KPS <80 was positively associated with preoperative crises (40). Englot et al. reported a reduced impact of preoperative crises in patients with cranial nerves deficits (7). However, studies on the frequency of symptoms have limits. Prospective studies are necessary to validate these potential predictors.
Postoperative predictors
Postoperative epilepsy/convulsions predictors are summarized in Table 2.
Table 2
Table 2 predictors of epilepsy/postoperative crises.
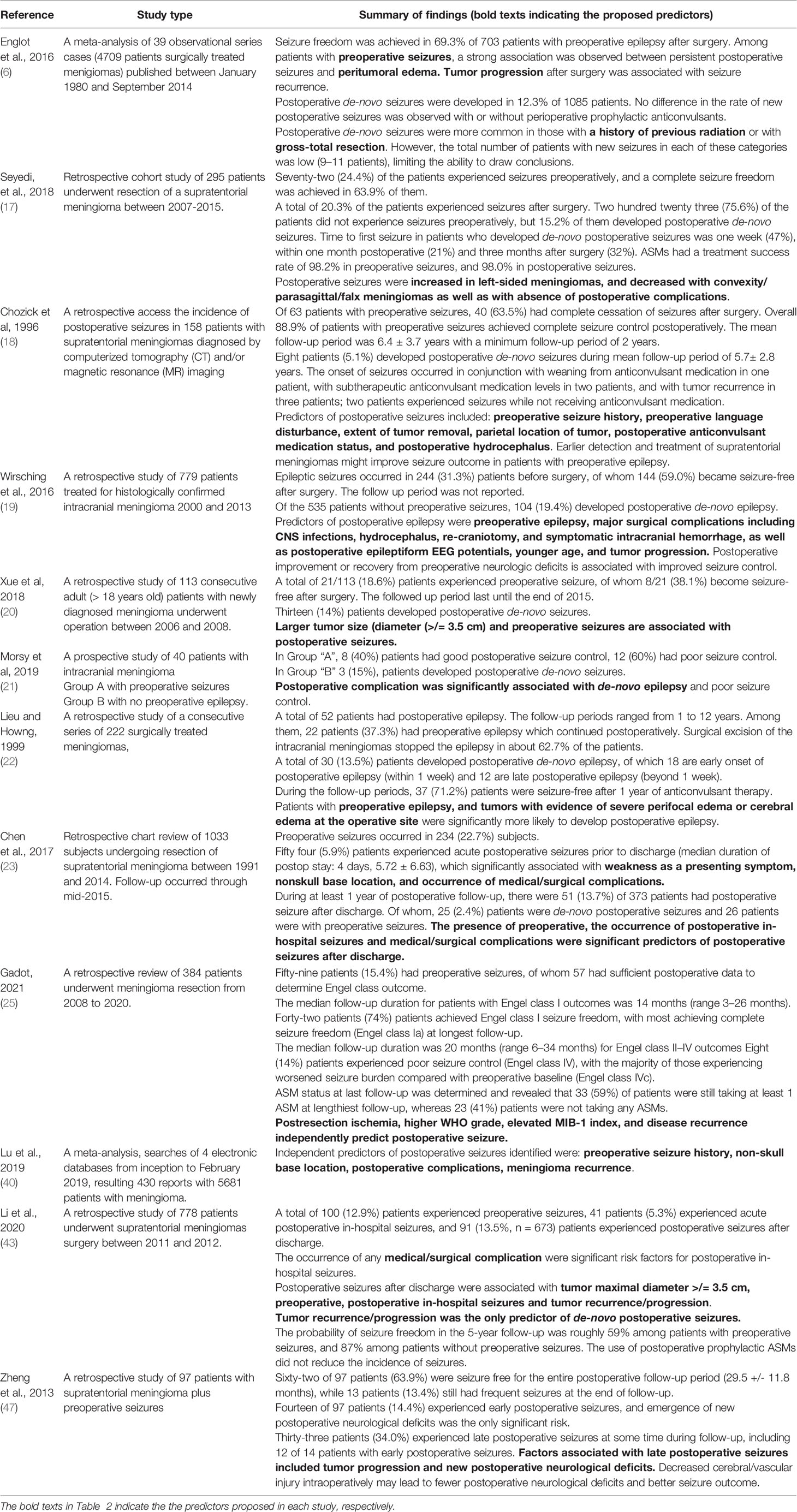
The International League against Epilepsy (LIG) defined acute postoperative crises as crises occurring within seven days of craniotomy (48). The late postoperative crisis is defined as an epilepsy crisis occurring beyond the first week of surgery (21, 49). In a retrospective study on 556 patients who underwent meningioma surgery, 74 patients presented postoperative crises, including 43% late crises (49). Certain studies have categorized postoperative crises in early, late crisis, in hospital and after leaving the hospital. The identification of possible predictors of postoperative crises can help guide the control of crises and to minimize the complications associated with the long -term use of ASM (13, 17, 20, 43, 47, 50).
The location of the tumor, its size, its grade, the damage to the motor zone and the KPS have all been studied as predictors of postoperative crises (21, 23). In a study, the occurrence of early crises in the hospital was associated with the involvement of the engine cortex, a postoperative KPS <70, postoperative complications and preoperative crises (43). It has been suggested that the decrease in the threshold and the increase in the sensitivity of the cortex during the immediate postoperative period are important factors to take into account, and that the use of ASMs can be justified during this period. The KPS <80 was an independent predictor of postoperative crises, with an almost three times higher risk of having preoperative crises (40). This still explains the impact of crises on quality of life. The lesions of the skull base were associated with a decrease in the incidence of preoperative crises, with an opposite trend and increased incidence in postoperative (40). Chen et al, in a study on 1033 patients, reported a decrease in the incidence of crises in the skull base lesions (20). The lesions of the skull base require greater brain retraction, which further increases brain edema (7, 51). Scott et al. Noted an association between meningioma on the left side and a greater risk of developing crises (52), with higher rates of postoperative crises reported in the left hemisphere (66.7%) compared to the right hemisphere (23.3%) (17). In a radiological study analyzing magnetic resonance imaging (MRI) structural in 3D of patients with meningiomas in order to identify the hot spots for crises, the results showed a high probability of crises when the lesion was located on the frontal lobe engine (44).
The preoperative crises were strong predictors of postoperative crises , in particular uncontrolled crises (13, 17, 20, 43). There is a contradiction in the literature concerning neurological deficits as a symptoms of presentation. In some studies, it was associated with a lower impact of preoperative crises (17, 20), and in others, it turned out to be significantly associated with postoperative crises before the exit (10, 19). During a univariate analysis, Chen et al. noted that a neurological deficit in the form of a new weakness, pneumonia, hematoma and infarction with edema was significantly associated with crises in the hospital. In their study, weakness was a predictive factor of crises in the hospital but not before or after the hospital leave (20). It is interesting to note that Wirsching et al. have found that postoperative improvement and recovery of preoperative neurological deficits were associated with a lower risk of postoperative crisis and better control (19).
Postoperative complications are independent predictors of postoperative crises (20). In the immediate postoperative period, the brain is more sensitive and its convulsion threshold is lowered (43). Any irritation of the very sensitive neocortex and probably still edematous can worsen crises immediately after surgery. A positive correlation has been established between postoperative complications such as hematoma, hydrocephalus, infection and edema (40). New permanent postoperative neurological deficits, especially in patients with vascular lesions, have significantly increased the risk of postoperative crises (47). Wirshing et al. Specified major surgical complications such as infections of the central nervous system, hydrocephalus, re-craniotomy and symptomatic intracranial hemorrhage as risk factors for postoperative seizures (19).
For crises after leaving the hospital, Li et al. identified the size of the tumor> 3.5 cm, preoperative crises and the progression of the tumor as important predictors (43). In the same study, postoperative complications were associated with acute postoperative crises, but no correlation with postoperative crises during long -term follow -up. In another study, surgical complications were associated with hospital crises and crises after leaving the hospital in patients who have never had crises (19, 53). Chen et al. did not find that tumor recurrence or subtotal resection were important predictors of postoperative crises (20). Englot et al. found a strong association between the deficits of the cranial nerves and the crises after leaving the hospital during a univariate analysis (7).
Surgical resection and freedom of epilepsy
Improving surgical techniques and the earlier diagnosis of meningioma had an impact on the extent of resection with favorable results . As indicated above, surgery eliminates epilepsy attacks in 70 % of patients, with rates ranging from 19 % to 90 % (7, 21). In some studies, the overall freedom of crisis over 5 years was 87% in patients with preoperative crises and 59% in naive crisis patients (4, 43). Lu et al. reported a rate of 30-40% postoperative crises in patients with a history of crisis before the operation and 10-15% in naive crisis patients (40). Komotar et al. have shown a significant influence of total gross resection on crisis rates (54). These reports support surgical intervention and cytoreduction in patients with persistent crises. On the other hand, it has been noted that the new postoperative crises were more frequent in patients who have undergone total coarse resection (46). A possible explanation is that a greater manipulation, dissection and retraction of the brain to achieve total coarse resection can cause cortical lesions, irritation, edema and crises. In a study, the resection of Grade I of Simpson was correlated with postoperative crises (39). Most of these lesions were convexity meningiomas, which are strongly correlated with epilepsy attacks. Therefore, the Simpson classification was not clinically relevant in this study. Similar results have been reported by Hess et al, without any statistical meaning being noted between the rank of Simpson and postoperative crises (35). Multiple studies have shown an association between crises and tumor recurrence/progression (23, 47). A postulated theory is that there is a possible reactivation of an anterior epileptogenic home or the formation of a new home with the recurrence of the tumor (40, 43). WHO grade i lesions have low recurrence rates and, in the event of coarse total resection, this can be a postoperative crisis protection factor (4, 5).
Most of the literature data report an absence of crises after a craniotomy and resection, and few studies deal with other treatment methods such as radiosurgery. Kondziolka et al. reported a case of mortality without more details (54). In Zada's study on 116 patients who have undergone treatment by gamma knife for meningioma, there was no crisis over the 75 months of follow -up (55). Pollack et al. pointed out a rate of 1.6 % of new or aggravated crises after radiosurgery (56). A decrease in freedom of crisis was reported after surgery in patients with intractable crises in preoperative (40).
Unaccompanied epilepsy management linked to meningioma; Medicines and epilepsy surgery
The American academy of neurology does not recommend the prophylactic use of ASMs in cases of newly diagnosed brain tumors. In our institution, we do not recommend obtaining a preoperative EEG to help determine whether the patient should be placed under ASM. However, some surgeons recommend the prophylactic use of ASMs in the immediate postoperative period in order to prevent Dé-Novo crises (57). In a study by Zheng et al. ASMs have reduced the risk of early postoperative crises (8, 58). ASMs can be used in patients with preoperative crises as a temporary measure until surgical resection. It is estimated that 40 % of patients whose crises were well controlled before surgery could be weaned ASM in the 27 postoperative months, and that only 22 % remained with intractable crises (8). For better selection of patients and better use of ASM postoperatively, the Stampe rating system was an attempt to help guide the treatment of epilepsy in patients with meningioma (19). They proposed a simple notation system including possible risk factors such as sensorimotor deficit, the progression of the tumor, age <55, a major surgical complication, preoperative crises, postoperative EEG and brain edema. However, the results were not statistically significant and required additional validation.
The evaluation of epilepsy surgery for further resection after having delimited the epileptogenic zone by intracranial EEG monitoring (grids, bands or stereotaxic electrodes), including peroperative ECOG, is the reference approach in level 4 epilepsy centers for patients with lesional epilepsy that did not respond to at least two ASM selected and dosed. The EEG can be useful to assess the recurrence of crises when weaning or stopping ASM. Several studies have suggested the systematic use of the POSE EEG to predict the recurrence of crises. In a study on 340 patients, the epileptiform discharge made it possible to predict postoperative crises, which pleads in favor of the systematic use of the EEG in postoperative (19). The peroperative mapping of ECOG and the resection of the secondary crisis home in the peritumoral cortex can increase the rates of absence of a postoperative crisis (23, 27). The postoperative EEG with epileptiform discharges is suggested as a predictor of the appearance of postoperative crises (19, 59). However, the American Academy of Neurology has published a practice directive in adult epileptic patients who obtained an absence of crisis (but not specifically for meningioma), the order of an EEG to detect inter -scileptive discharges is not useful to guide the decision to prosecute the ASM. However, this approach is more reliable in pediatric patients. An epileptiform potential on EEG in pediatric patients increases the risk of crisis recurrence (60).
In our center, we evaluate each patient with epilepsy linked to meningioma, in particular patients who continue to have uncontrolled postoperative seizures with the implementation of deep stereotaxic electrodes (S-EEG) or by under-dural/band underline electrodes, and in cases where functional cartography is essential to exclude the implication of the cortex epileptogenic. S-EEG is a safer option for patients who must undergo a second surgical intervention, knowing that the complications of the previous intervention, such as adhesions, infections, bleeding, etc. are likely to be a problem. Rise functional mapping can also be carried out by EEG-S in comparison with the detailed functional map carried out by grid or band electrodes. In areas where safer resection is possible outside the eloquent cortex, EEG-S is useful for encompassing the edges of the lesion and to reach distant areas of interest, such as mesial temporal structures, in order to exclude a double pathology.
The following case illustrates our own experience in the management of postoperative crises after a resection of meningioma. A left -handed 36 -year -old man who underwent a median left -wing parasagittal meningiomal (6 x 7 cm) and has developed recent appearance seizures 8 to 10 months after the operation. His MRI 3 months after the operation showed a complete resection of the tumor. About 11 months after resection, he developed his first crisis "Tonico-Clonique". It started with numbness, weakness and tingling on the right side of his back, descending towards the middle of his spine. It was then put under lamotrigine, but continued to have repeated crises which began with the same feelings of swarming to the spine, associated with abnormal sensations of butterfly in the abdomen, to finally lead to movements of trembling of the right foot, with propagation to the right arm. Due to the development of drug-resistant epilepsy (ERD), including lamotrigine, lacosamide and Levetiracetam, he underwent additional surgical evaluation of epilepsy, including an EEG video of the scalp and intracranial EEG monitoring with stereotaxic intracranial S-EEG monitoring, which has better Epileptogenic zone in the central and paracentral frontal channels left behind the posterior and mesial margins of the surgical cavity, with probably an earlier appearance on the mesial surface on the left side of the interhemispherical crack given the early implication of the right foot (Figure 1). About 25 months after initial surgery, he underwent a second left frontal craniotomy programmed for the resection of epileptogenic foci. He continued to take anticonvulsant drugs after the operation and reported an improvement in the frequency of crises. Since he underwent his second surgery, there has been a significant reduction in the frequency of crises, which has gone from twice a week to twice a year, of the conscious focal type triggered by the reduction of drugs or the consumption of alcohol.
MRI 1
MRI 1 Circles represent the active inter -scileptive discharges (irritative areas). The stars represent the first contacts involved at the start of the epileptic episode. The dotted lines represent the proposed resection area.
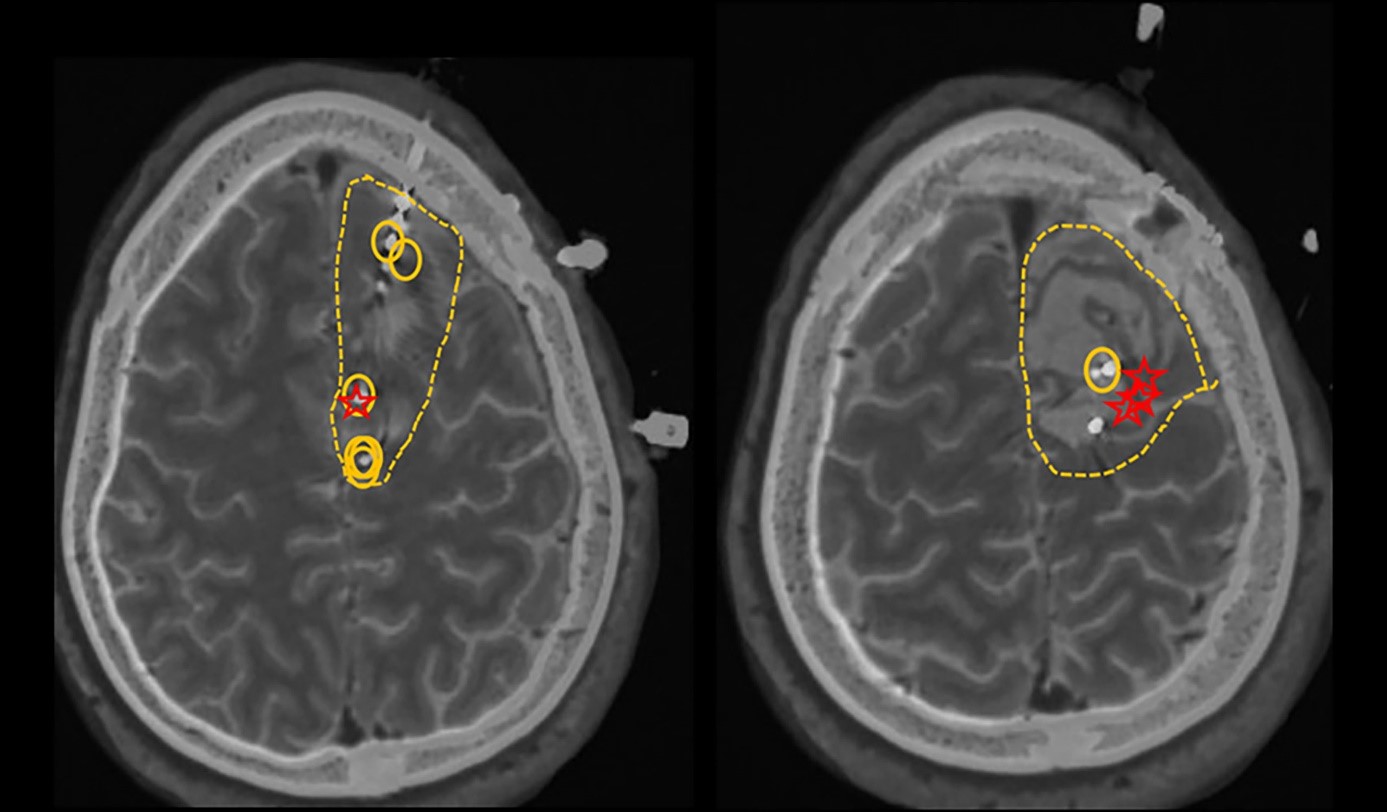
This case report is used as an example to show the complexity of the management of a patient with meningioma having undergone a craniotomy. A separate project approved by the IRB will be carried out to retrospectively analyze the success rate of such procedures in our center.
Regarding the medical treatment of primary brain tumors (TCP) in general, there are no solid and randomized studies to support the choice of ASMs. Several factors should be taken into account, including sex, age, cost, profession, cognition, common side effects of drugs, the neurological base linked to tumor/surgery (in order to avoid the addition of the undesirable effects of drugs), pharmacokinetics of drugs, drug interactions, efficiency and comorbidities. It is also necessary to take into account the interaction with chemotherapy treatments and the effect of radiation on the brain. Certain types of tumors (such as low-grade tumors) are known to be resistant to ASM treatment due to several hypotheses such as the intrinsic severity of the underlying mechanism of epileptogenicity, the altered expression of the molecules on which the ASM act or the modification of the expression of the transporters at the level of the hemato-encephalic barrier limiting the penetration of the drug epileptogenic (61). The most recent ASMs (oxcarbazepine, topiramate, lamotrigine, levetiracetam, zonisamide and lacosamide) have better tolerance and better efficiency due to different aspects, including their property of not indzyme, their limited interaction between drugs, their pure kidney excretion and their lesser side effects. Old generation ASMs such as carbamazepine, phenytoin and phenobarbital are falling into disuse because of their strong protein bond, drug interactions and the induction of the hepatic P-450. The undesirable effects of ASMs are reported as higher in PBT patients than in the general population of epileptics (24% vs 0.5-12%) (57). In PBTS, the undesirable effects of the ASMS directed towards the brain function such as the executive function, the attention of attention, the cognitive function are six times higher than the undesirable effects linked to the irradiation of the brain (62). Overall, the best risk-benefit report for the choice of ASM to be used is based on the doctor's judgment. It is very important to mention that treatment must be started after a single crisis. According to the recommendations of the American Academy of Neurology (ANA), it is not necessary to administer a prophylactic treatment by ASM to patients with brain tumors without a history of crises. It is also suggested that the gradual decrease and the stop of ASM after the first postoperative week are appropriate in the absence of crisis history (57). In summary, the strategy of selecting drugs for the management of the BTR must promote parenteral administration drugs, the ASMs which do not require slow titration, and must avoid enzymatic inductors. If monotherapy fails, you must consider combined therapy, poor compliance, repeated surgery and tumor recurrence/progression.
The efficiency of each ASM is very variable: oxcarbazepine in monotherapy: 62.9 %; Topiramate in monotherapy: 55.6 %; Gabapentine, pregabalin, tiagabine, zonisamide in extra treatment: 27.4 to 100 %; Levetiracetam in monotherapy and extra treatment: 47.4% at 88%; Lacosamide as an extra medication with 42.9% (63). Levetiracetam and valproic acid are the most studied drugs in epilepsy linked to tumors. Levétiracétam was studied compared to valproic acid and the failure of the treatment of crises in glioblastoma was 33% against 50%, perhaps because of its tolerance and its property to strengthen the inhibition of methylguanine-add Methyltransferase by P53 in this population of patients (64). The most attractive factors for the popularity of the Levetiracetam are its good tolerance, its ease of use without the need for titration, the lack of interaction with other ASM, its hepatic non-metabolization by the CYP450, and therefore the lack of interaction with certain chemotherapy drugs used in certain cases of BTR, and finally good insurance coverage.
In a recent survey published on ASM preferences of prescriptions among European neuro-oncologists, Levetiracetam is considered to be the first choice for patients with brain tumors, with the highest presumed efficiency and the weakest adverse effects (65). ASMs differ by their pharmacokinetics, their therapeutic efficiency and their side effects, which were examined in detail by Maschio (63).
The care of crises must go beyond pharmacological options . Untreated crises can expose patients at a risk of catastrophic outcome, such as unexpected sudden death in epileptic patients. In addition, crises can have a negative impact on the lifestyle of patients, especially on work, employment, education and driving. The risk of physical injury or death is not limited to the driver and passengers, but applies to pedestrians and people in other vehicles. The different American states have different laws to determine which group of epileptic patients can drive. The crises can cause other physical injuries. Patients with refractory epilepsy must be treated in tertiary centers where they can benefit from medical, social and behavioral support and, above all, an evaluation for epileptic surgery.
Future prospects
Despite the progress made in understanding the pathophysiological mechanisms and the management of epilepsy linked to meningiomas, important gaps in knowledge remain .
Among the relevant questions, let us quote: " What are the patients most exposed to the risk of epileptic crises? »And» When to start ASM and for how long? ".
The risk of persistent postoperative crises highlights the need to continue research on crises in patients with meningioma. The arbitrary and long -term use of ASMs in patients with meningioma highlights the importance of directives for appropriate selection of patients. Thus, randomized prospective trials are necessary to guide the selection and prescription of ASMs. Stop 'EM is a controlled randomized trial in progress, the end date of which is set at September 2027 (66). It aims to determine the need to use postoperative ASM in patients who have never had an epileptic crisis. The main objectives of the study are to determine the effectiveness of the levetiracetam in crisis prevention over a period of 12 months after surgery, the effect of starting the levetiracetam on the ability to resume conduct, quality of life and cost-effectiveness.
Conclusion
Understanding and predicting epilepsy seizures related to meningioma can help guide crisis control and better determine patients at risk before and after surgery. The current medical literature provides limited data for the prediction of postoperative crises and optimal management of patients with epilepsy linked to meningioma.
With regard to the cohort of patients with meningioma and undergoing surgery, stratified according to the state of preoperative crisis compared to the postoperative crisis state, it is logical to identify four different groups:
- without crisis in relation to the state of crisis,
- with crisis in relation to the state of crisis,
- without crisis in relation to the state of crisis
- And with crisis in relation to the state of crisis.
Future efforts to stratify patients in these four groups, including drugs alone, surgery/interventions alone, drugs + surgery/interventions , will make it possible to predict the result of surgery and to treat patients optimally with the most effective methods.
Contributions from authors
Re, HT, LH, WB and FB contributed to the design and design of the study. RE wrote the first version of the manuscript. AA has written sections of the manuscript. HT, LH, AA, WB and FB contributed to the revision of the manuscript. All the authors approved the submissive version.
Conflict of interest
The authors declare that research has been carried out in the absence of any commercial or financial relationship which could be interpreted as a potential conflict of interest.
Publisher's note
All the claims expressed in this article only engage the authors and do not necessarily represent those of their affiliated organizations, nor those of the publisher, editors and revisers. Any product evaluated in this article, or any claim made by its manufacturer, is neither guaranteed nor approved by the publisher.
Thanks
This study was supported by the subsidy (n ° 19140900105) of the Shanghai Science and Technology Committee and by the Research Fund of the Neurosurgery Department of the University of Loma Linda.
References
- Wiemels J, Wrensch M, Claus EB. Epidemiology and Etiology of Meningioma. J Neurooncol (2010) 99 (3): 307–14. DOI: 10.1007/S11060-010-0386-3
Pubmed abstract | Crossref Full Text | Google Scholar
- Porter Kr, McCarthy BJ, Freels S, Kim Y, Davis FG. Prevalence Estimates for Primary Brain Tumors in the United States by Age, Gender, Behavior, and Histology. Neuro Oncol (2010) 12 (6): 520–7. DOI: 10.1093/Neuonc/NOP066
Pubmed abstract | Crossref Full Text | Google Scholar
- Ostrom Qt, Gittleman H, Fulop J, Liu M, BLANDA R, Kromer C, et al. CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2008-2012. Neuro Oncol (2015) 17 Suppl 4: IV1–IV62. DOI: 10.1093/Neuonc/Nov189
Pubmed abstract | Crossref Full Text | Google Scholar
- Sughrue Me, Kane AJ, Shangari G, Rutkowski MJ, McDermott MW, Berger MS, et al. The lift of Simpson Grade I and II Resection in Modern Neurosurgical Treatment of World Health Organization Grade I Meningiomas. J Neurosurg (2010) 113 (5): 1029–35. DOI: 10,3171/2010.3.JNS091971
Pubmed abstract | Crossref Full Text | Google Scholar
- Hasseleid BF, MELING TR, RONNING P, Scheie D, HelSeth E. Surgery for Convexity Meningioma: Simpson Grade I Resection As the Goal: Clinical Article. J Neurosurg (2012) 117 (6): 999–1006. DOI: 10.3171/2012.9.JNS12294
Pubmed abstract | Crossref Full Text | Google Scholar
- Erturk Cetin O, Isler C, Uzan M, Ozkara C. Epilepsy-Related Brain Tumors. Seizure (2017) 44: 93–7. DOI: 10.1016/J. SEIZURE.2016.12.012
Pubmed abstract | Crossref Full Text | Google Scholar
- Englot DJ, Magill St, Han SJ, Chang EF, Berger MS, McDermott MW. Seizures in supratentorial meningioma: a systematic review and meta-analysis. J Neurosurg (2016) 124 (6): 1552–61. DOI: 10.3171/2015.4.JNS142742
Pubmed abstract | Crossref Full Text | Google Scholar
- Chaichana KL, Pendleton C, Zaidi H, Olivi A, Weingart JD, Gallia GL, et al. Seizure Control for patients undergoing meningioma surgery. World Neurosurg (2013) 79 (3-4): 515–24. DOI: 10.1016/J.WNEU.2012.02.051
Pubmed abstract | Crossref Full Text | Google Scholar
- Van Breemen MS, Wilms EB, Vecht CJ. Seizure Control in Brain Tumors. Handb Clin Neurol (2012) 104: 381–9. DOI: 10.1016/B978-0-444-52138-5.00026-8
Pubmed abstract | Crossref Full Text | Google Scholar
- Bauer R, ORTLER M, SEIZZ-ROSENAGEN M, MAIER R, Anton JV, UNTERBERGER I. TREATMENT OF EPILEPTIC SEIZURES IN BRAIN TUMORS: A Critical Review. Neurosurg Rev (2014) 37 (3): 381–8: discussion 8. DOI: 10.1007/S10143-014-0538-6
Pubmed abstract | Crossref Full Text | Google Scholar
- Taphoorn MJ, Klein M. Cognitive Deficits in Adult Patients With Brain Tumours. Lancet Neurol (2004) 3 (3): 159–68. DOI: 10.1016/S1474-4422 (04) 00680-5
Pubmed abstract | Crossref Full Text | Google Scholar
- Gilliam F, Kuzniecky R, Faught E, Black L, Carpenter G, Schrodt R. Patient-Validated Content of Epilepsy-Specific Quality-of-Life Measurement. Epilepsia (1997) 38 (2): 233–6. DOI: 10.1111/J.1528-1157.1997.TB01102.x
Pubmed abstract | Crossref Full Text | Google Scholar
- Chaichana KL, Parker SL, Olivi A, Quinones-Hinojosa A. Long-Term seizure outcomes in Adult patients undergoing Primary Resection of Malignant Brain Astrocytomas. Clinical article. J Neurosurg (2009) 111 (2): 282–92. DOI: 10.3171/29.2.jns081132
Pubmed abstract | Crossref Full Text | Google Scholar
- Harden cl. The Co-Morbidity of Depression and Epilepsy: Epidemiology, Etiology, and Treatment. Neurology (2002) 59 (6 Suppl 4): S48–55. DOI: 10.1212/WNL.59.6_SUPPL_4.S48
Pubmed abstract | Crossref Full Text | Google Scholar
- Cramer JA, Mintzer S, Wheless J, Mattson HR. Adverse Effects of Antiepileptic Drugs: A Brief Overview of Important. Rev Neurother Expert (2010) 10 (6): 885–91. DOI: 10.1586/ERN.10.71
Pubmed abstract | Crossref Full Text | Google Scholar
- Hamasaki t, yamada k, kuratsu J. seizures as a presenting symptom in neurosurgical patients: a retrospective single-institution analysis. Clin Neurol Neurosurg (2013) 115 (11): 2336–40. DOI: 10.1016/J.Clineuro. 2013.08.016
Pubmed abstract | Crossref Full Text | Google Scholar
- Seyedi JF, Pedersen CB, Poulsen fr. Risk of Seizures Before and After Neurosurgical Treatment of Intracranial Meningiomas. Clin neurol neurosurg (2018) 165: 60–6. DOI: 10.1016/J.Clineuro. 2018.01.002
Pubmed abstract | Crossref Full Text | Google Scholar
- Chozick BS, Reinert SE, Greenblatt SH. Incidence of seizures after surgery for supratentorial meningiomas: a modern analysis. J Neurosurg (1996) 84 (3): 382–6. DOI: 10.3171/JNS.1996.84.3.0382
Pubmed abstract | Crossref Full Text | Google Scholar
- Wirsching HG, Morel C, GMUR C, Neidert MC, Baumann CR, Valavanis A, et al. Predicting outcome of epilepsy after meningioma resection. Neuro Oncol (2016) 18 (7): 1002–10. Doi: 10.1093/Neuonc/Nov303
Pubmed abstract | Crossref Full Text | Google Scholar
- Chen WC, Magill St, Englot DJ, Baal JD, Wagle S, Rick JW, et al. Factors Associated with pre- and postoperative seizures in 1033 patients undergoing supratentorial meningioma resection. Neurosurgery (2017) 81 (2): 297–306. Doi: 10.1093/Neuros/NYX001
Pubmed abstract | Crossref Full Text | Google Scholar
- Place as, Howng SL. Intracranial meningiomas and epilepsy: incidence, prognosis and influencing factors. Epilepsy res (2000) 38 (1): 45–52. DOI: 10.1016/S0920-1211 (99) 00066-2
Pubmed abstract | Crossref Full Text | Google Scholar
- Morsy MM, El-Saadany WF, Moussa WM, Sultan AE. Predictive factors for seizures accompanying intracranial meningiomas. Asian J Neurosurg (2019) 14 (2): 403–9. Doi: 10.4103/AJNS.AJNS_152_18
Pubmed abstract | Crossref Full Text | Google Scholar
- Xue H, Sveinson O, Bartek J Jr., Forander P, Skyrman S, Kihlstrom L, et al. Long-Term Control and Predictors of Seizures In Intracranial Meningioma Surgery: A Population-Based Study. Acta Neurochir (Wien) (2018) 160 (3): 589–96. DOI: 10.1007/S00701-017-3434-3
Pubmed abstract | Crossref Full Text | Google Scholar
- Schaller B. Brain Tumor and Seizures: Pathophysiology and its implications for Treatment Revisited (Epilepsia 2003; 44: 1223-1232). Epilepsia (2006) 47 (3): 661; discussion. DOI: 10.1111/J.1528-1167.2006.00484_1.x
Crossref Full Text | Google Scholar
- Gadot R, Khan AB, Patel R, Goethe E, Shetty A, Hadley CC, et al. PREDICTORS OF POSTOPERATIVE SEIZURE OUTCOME IN SUPRATORIAL MENINGIOMA. J neurosurg (2021), 1–10. DOI: 10.3171/2021.9.JNS211738
Pubmed abstract | Crossref Full Text | Google Scholar
- Shamji MF, money-shamji EC, Benoit BG. Brain Tumors and Epilepsy: Pathophysiology of Peritumoral Changes. Neurosurg Rev (2009) 32 (3): 275–84; Discussion 84-6. DOI: 10.1007/S10143-009-0191-7
Pubmed abstract | Crossref Full Text | Google Scholar
- Fang S, Zhan Y, Xie Yf, Shi Q, Dan W. Predictive Value of Electrocorticography for Postoperative Epilepsy in Patients with supratentorial meningioma. J Clin Neurosci (2013) 20 (1): 112–6. DOI: 10.1016/J.JOCN.2012.02.021
Pubmed abstract | Crossref Full Text | Google Scholar
- Van Diessen E, Diederen SJ, Braun KP, Jansen Fe, Stam CJ. Functional and structural Brain Networks in Epilepsy: What Have We Learned? Epilepsia (2013) 54 (11): 1855–65. Doi: 10.1111/EPI.12350
Pubmed abstract | Crossref Full Text | Google Scholar
- Mehta n, bhagwati s, Parulekar G. Meningiomas in Children: A Study of 18 boxes. J Pediatr Neurosci (2009) 4 (2): 61–5. DOI: 10.4103/1817-1745.57322
Pubmed abstract | Crossref Full Text | Google Scholar
- Simis a, worse from Aguiar pH, leite cc, santana pa jr., rosemberg s, teixeira mj. Peritumoral Brain Edema in Benign Meningiomas: Correlation with Clinical, Radiologic, and Surgical Factors and Possible Role on Reverse. Surg Neurol (2008) 70 (5): 471–7; Discussion 7. DOI: 10.1016/J.Surneu. 2008.03.006
Pubmed abstract | Crossref Full Text | Google Scholar
- Lobato RD, Alday R, Gomez PA, Rivas JJ, Dominguez J, Cabrera A, et al. Brain edema in Patients with intracranial meningioma. Correlation Between Clinical, Radiological, and Histological Factors and the Presence and Intensity of Edema. Acta Neurochir (Wien) (1996) 138 (5): 485–93; Discussion 93-4. DOI: 10.1007/BF0141166
Pubmed abstract | Crossref Full Text | Google Scholar
- Kawaguchi T, Kameyama S, Tanaka R. Peritumoral Edema and Seizure in Patients With Cerebral Convexity and Parasagittal Meningiomas. Neurol Med Chir (Tokyo) (1996) 36 (8): 568–73; Discussion 73-4. DOI: 10.2176/NMC.36.568
Pubmed abstract | Crossref Full Text | Google Scholar
- Yoshioka H, Hama S, Taniguchi E, Sugiyama K, Arita K, Kurisu K. Peritumoral Brain Edema Associated With Meningioma: Influence of Vascular Endothelial Growth Factor Expression and Vascular Blood Supply. Cancer (1999) 85 (4): 936–44. Doi: 10.1002/(SICI) 1097-0142 (19990215) 85: 4 <936 :: AID-CNCR23> 3.0.co; 2-J
Pubmed abstract | Crossref Full Text | Google Scholar
- Pistolesi s, Fontanini G, Camacci T, of Ieso K, Boldrini L, Lupi G, et al. Meningioma-associated brain edema: The Role of Angiogenic Factors and Pial Blood Supply. J Neurooncol (2002) 60 (2): 159–64. DOI: 10.1023/A: 1020624119944
Pubmed abstract | Crossref Full Text | Google Scholar
- Hess K, Spille DC, Adeli A, Sporns Pb, Brokinkel C, Grauer O, et al. Brain Invasion and the Risk of Seizures in Patients With Meningioma. J Neurosurg (2018) 130 (3): 789–96. DOI: 10,3171/2017.11.jns172265
Pubmed abstract | Crossref Full Text | Google Scholar
- From Vries J, Wakhloo AK. Cerebral Odema Associated With Who-I, Who-II, and Who-Iii-Meningiomas: Correlation of Clinical, Compudé Tomographic, Operative and Histological Findings. Acta Neurochir (Wien) (1993) 125 (1-4): 34–40. DOI: 10.1007/BF01401825
Pubmed abstract | Crossref Full Text | Google Scholar
- Chernov MF, Kasuya H, Nakaya K, Kato K, Ono Y, Yoshida S, et al. (1) H-MRS OF INTRACRANIAL MENINGIOMAS: What It can add to known clinical and mri predictors of the histopathological and biological characteristics of the tumor? Clin neurol neurosurg (2011) 113 (3): 202–12. DOI: 10.1016/J.Clineuro.2010.11.008
Pubmed abstract | Crossref Full Text | Google Scholar
- Spille DC, Hess K, Sauerland C, Sanai N, Stummer W, Paulus W, et al. Brain invasion in meningiomas: incidence and correlations with clinical variables and prognosis. World Neurosurg (2016) 93: 346–54. DOI: 10.1016/J.WNEU.2016.06.055
Pubmed abstract | Crossref Full Text | Google Scholar
- Islim AI, McKeever S, Kusu-Okar Te, Jenkinson MD. The Role of Prophylactic Antiepileptic Drugs for Seizure Prophylaxis in Meningioma Surgery: A Systematic Review. J Clin Neurosci (2017) 43: 47–53. DOI: 10.1016/J.JOCN.2017.05.020
Pubmed abstract | Crossref Full Text | Google Scholar
- Lu VM, Wahood W, Akinduro Oo, Parney If, Quinones-Hinojosa A, Chaichana Kl. Four independent predictors of postoperative seizures after meningioma surgery: a meta-analysis. World Neurosurg (2019) 130: 537–45 E3. DOI: 10.1016/J.WNEU.2019.06.063
Pubmed abstract | Crossref Full Text | Google Scholar
- Wilson Ta, Huang L, Ramanathan D, Lopez-Gonzalez M, Pillai P, de los Reyes K, et al. Review of Atypical and Anaplastic Meningiomas: Classification, Molecular Biology, and Management. Front Oncol (2020) 10: 565582. DOI: 10.3389/FONC. 2010.565582
Pubmed abstract | Crossref Full Text | Google Scholar
- Tsuji m, shinomiya s, inoue r, Sato K. Prospective Study of Postoperative Seizure In Intracranial Meningioma. JPN J Psychiatry Neurol (1993) 47 (2): 331–4. DOI: 10.1111/J.1440-1819.1993.TB02094.x
Pubmed abstract | Crossref Full Text | Google Scholar
- Li X, Wang C, Lin Z, Zhao M, Ren X, Zhang X, et al. Risk Factors and Control of Seizures in 778 Chinese Patients Initial Undergo Resection of Supratent Meningiomas. Neurosurg Rev (2020) 43 (2): 597–608. DOI: 10.1007/S10143-019-01085-5
Pubmed abstract | Crossref Full Text | Google Scholar
- Hamasaki T, Yamada K, Yano S, Nakamura H, Makino K, Hide T, et al. Higher Incidence of Epilepsy in Meningiomas Located On The Premotor Cortex: A Voxel-Wise Statistical Analysis. Acta Neurochir (Wien) (2012) 154 (12): 2241–9. DOI: 10.1007/S00701-012-1511-1
Pubmed abstract | Crossref Full Text | Google Scholar
- McGovern SL, Aldape KD, Munsell MF, Mahajan A, Demonte F, Woo Sy. A comparison of World Health Organization Tumor Grades At Patients in Patients With Non-Skull Base and Skull Base Meningiomas. J Neurosurg (2010) 112 (5): 925–33. DOI: 10,3171/29.9.jns09617
Pubmed abstract | Crossref Full Text | Google Scholar
- Baumgarten P, Sarlak M, Baumgarten G, Marquardt G, Seifert V, Strzelczyk A, et al. Focused Review on Seizures Caused by Meningiomas. Epilepsy Behav (2018) 88: 146–51. DOI: 10.1016/J.Yebeh. 2018.09.002
Pubmed abstract | Crossref Full Text | Google Scholar
- Zheng Z, Chen P, Fu W, Zhu J, Zhang H, Shi J, et al. Early and postoperative late seizure outcoma in 97 patients with supratentorial meningioma and preoperative seizures: a retrospective study. J Neurooncol (2013) 114 (1): 101–9. DOI: 10.1007/S11060-013-1156-9
Pubmed abstract | Crossref Full Text | Google Scholar
- Beghi E, Carpio A, Forsgren L, Hesdorffer DC, Malmgren K, Sander JW, et al. Recommendation for a definition of acute symptomatic seizure. Epilepsia (2010) 51 (4): 671–5. DOI: 10.1111/J.1528-1167.2009.02285.x
Pubmed abstract | Crossref Full Text | Google Scholar
- Baumgarten P, Sarlak M, Monden D, Spyrantis A, Bernatz S, Gessler F, et al. Early and postoperative late seizures in meningioma patients and prediction by a recent scoring system. Cancers (Basel) (2021) 13 (3): 450. DOI: 10.3390/Cancers13030450
Pubmed abstract | Crossref Full Text | Google Scholar
- Joiner EF, Youngerman Be, Hudson TS, Yang J, Welch MR, McKhann GM, et al. Effectiveness of Perioperative Antiepileptic Drug Prophylaxis for Early and Late Seizures Following Oncologic Neurosurgery: A Meta-Analysis. J Neurosurg (2018) 130 (4): 1–9. DOI: 10.3171/2017.10.JNS172236
Pubmed abstract | Crossref Full Text | Google Scholar
- Raza SM, Gallia GL, Brem H, Weingart JD, Long DM, Olivi A. Perioperative and long-Term OUTCOMES from the management of parasagittal meningiomas invading the superior sagittal sinus. Neurosurgery (2010) 67 (4): 885–93; Discussion 93. DOI: 10.1227/Neu. 0B013E3181EF2A18
Pubmed abstract | Crossref Full Text | Google Scholar
- Scott DF. LEFT AND RIGHT CEREBRAL HEMISPHERE DIFFERENCES IN THE OPPERY OF EPILEPSY. BR J Med Psychol (1985) 58 (PT 2): 189–92. DOI: 10.1111/J.2044-8341.1985.TB02633.x
Pubmed abstract | Crossref Full Text | Google Scholar
- Islim AI, Ali A, Bagchi A, Ahmad Mu, Mills SJ, ChavreDakis E, et al. Postoperative seizures in meningioma patients: improving patient selection for antiepileptic drug therapy. J Neurooncol (2018) 140 (1): 123–34. DOI: 10.1007/S11060-018-2941-2
Pubmed abstract | Crossref Full Text | Google Scholar
- KOMOTAR RJ, RAPER DM, Starke RM, IIORGULESCU JB, GUTIN PH. Prophylactic antiepileptic drug therapy in patients undergoing supratentorial meningioma resection: a systematic analysis of efficient. J Neurosurg (2011) 115 (3): 483–90. DOI: 10.3171/2011.4.JNS101585
Pubmed abstract | Crossref Full Text | Google Scholar
- Zada G, Pagnini PG, Yu C, Erickson KT, Hirschbein J, Zelman V, et al. Long-term outcomes and patterns of tumor progression after gamma knife radiosurgery for benign meningiomas. Neurosurgery (2010) 67 (2): 322–8; Discussion 8-9. DOI: 10.1227/01.NEU.0000371974.88873.15
Pubmed abstract | Crossref Full Text | Google Scholar
- Pollock Be, Stafford SL, Link MJ, Yi Graces, Fote RL. Single-Fraction Radiosurgery for Presumed Intracranial Meningiomas: efficient and complications from 2-Year experience. Int J Radiat Oncol Biol Phys (2012) 83 (5): 1414–8. DOI: 10.1016/J.IJROBP.2011.10.033
Pubmed abstract | Crossref Full Text | Google Scholar
- Glantz MJ, Cole BF, Forsyth PA, Recht LD, Wen Py, Chamberlain Mc, et al. PERMETER PRACTICE: Anticonvulser prophylaxis in Patients with newly diagnosed brain tumors. Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology (2000) 54 (10): 1886–93. DOI: 10.1212/WNL. 54.10.1886
Pubmed abstract | Crossref Full Text | Google Scholar
- Zhang B, Zhao G, Yang HF, Wang D, Yu JL, Huang Hy. Assessment of Risk Factors for Early Seizures Following Surgery for Meningiomas Using Logistic Regression Analysis. J int Med Res (2011) 39 (5): 1728–35. DOI: 10.1177/147323001103900515
Pubmed abstract | Crossref Full Text | Google Scholar
- Harward SC, Rolston JD, Englot DJ. Seizures in meningioma. Handb Clin Neurol (2020) 170: 187–200. DOI: 10.1016/B978-0-12-822198-3.00053-7
Pubmed abstract | Crossref Full Text | Google Scholar
- Gloss D, Pargeon K, Pack A, Varma J, French Ja, Tolchin B, et al. Antiserizure Medication Withdrawal in Seizure-Free Patients: Practice Advisory Update Summary: Report of the Aan Guideline Subcommittee. Neurology (2021) 97 (23): 1072–81. DOI: 10.1212/WNL.00000000000129444
Pubmed abstract | Crossref Full Text | Google Scholar
- Guerrini R, Rosati A, Giordano F, Genitori L, Barba C. The Medical and Surgical Treatment of Tumoral Seizures: Current and Future Perspectives. Epilepsia (2013) 54: 84–90. Doi: 10.1111/EPI.12450
Pubmed abstract | Crossref Full Text | Google Scholar
- Klein M. Neurocognitive Functioning in Adult Who Grade II Glimas: Impact of Old and New Treatment Modalities. Neuro Oncol (2012) 14: 17–24. DOI: 10.1093/Neuonc/NOS161
Crossref Full Text | Google Scholar
- Maschio M, Dinapoli L, Sperati F, Pace A, Fabi A, Vidiri A, et al. Effect of pregabalin Add-on Treatment on Seizure Control, Quality of Life, and anxiety in Patients with Brain Tumour-Related Epilepsy: A Pilot Study. Epileptic Disord (2012) 14 (4): 388–97. DOI: 10.1684/EPD.2012.0542
Pubmed abstract | Crossref Full Text | Google Scholar
- van der Meer pb, dirven l, fiocco m, your mj, kouwenhoven McM, van den bent mj, et al. FIRST-Line Antiepileptic Drug Treatment in Gloma Patients with Epilepsy: Levetiracetam vs valproic acid. Epilepsia (2021) 62 (5): 1119–29. Doi: 10.1111/EPI.16880
Pubmed abstract | Crossref Full Text | Google Scholar
- van der meer pb, dirven l, van den bent mj, prelyus m, taphoorn mjb, rudá r, et al. Prescription preferences of antiepileptic drugs in brain tumor patients: an international survey among eano members. Neuro-oncol PRACT (2021) 9 (2): 105–13. Doi: 10.1093/NOP/NPAB059
Crossref Full Text | Google Scholar
- Jenkinson M, Helmy A, Huckey H, Mills S, Grant R, Hughes D, et al. Surgeons Trial of Prophylaxis for Epilepsy in Seizure Naïve Patients With Meningioma: A Randomized Controlled Trial (Stop 'EM). Neuro-oncol PRACT (2020) 22 (Supplement_2): II195. DOI: 10.1093/Neuonc/Noaa215.815
Crossref Full Text | Google Scholar





