Association between hormonal contraception and the location of meningiomas in Indonesian patients
Rusdy Ghazali Malueka 1 , Rachmat Andi Hartanto 2 , Nurhuda Hendra Setyawan 3 , Dyajeng Noor Firdaus Fauzi 1 , Khoironi Rachmad Damarjati 1 , Alfian Rismawan 1 , Maria Alethea Septianasti 1 , Adiguno Suryo Wicaksono 2 , Kusumo Dananjoyo 1 , Endro Basuki 2 , Ahmad Asmedi 1 , Ery Kus Dwianingsh 4 *
Summary
Context : meningioma is the most frequent primary intracranial tumor. Previous studies have shown a possible association between the use of hormonal contraceptives and the location of meningioma. This study therefore aimed to analyze the association between the history of the use of hormonal contraceptives and the location of meningiomas in the Indonesian population.
Methods : in total, 99 patients with meningioma confirmed histologically and admitted to the Dr. Sardjito general hospital in Yogyakarta, Indonesia, were included in this study. Hormonal contraception data and other variables were collected from medical records. The location of meningiomas was determined from magnetic resonance imaging (MRI) or computer tomography (CT) of the brain before surgery.
Results : Sixty-two (72.7 %) patients had history of hormonal contraception. The subjects consist of 83 (83.8%) OMS grade I and 16 (16.2%) tumors of Grade II and III of WHO. A total of 57 (57.6 %) tumors were located in the Spheno-Orbital region. We have found a significant association between the use of hormonal contraceptives and the location of meningiomas in the spheno-orbital region (Odds ratio (gold) 2.573, p = 0.038). As a result, patients in the hormonal contraception group had a greater visual impairment (p = 0.044).
Conclusion : The use of hormonal contraception is associated with the location of meningiomas in the Spheno-Orbital region.
Keywords : meningioma- Location of meningioma- hormonal contraception- Indonesian population.
Asian PAC J Cancer Prev, 23 (3), 1047-1051
Introduction
Meningioma is the most common primary intracranial tumor which is born from meningothelial cells of the arachnoid layer. It represents approximately 36 % of primary intracranial tumors, 78.9 % of which are located in intracranial and 4.2 % in the spine (Ostrom et al., 2018). According to the central register of brain tumors in the United States, the meningioma incidence rate was 8.58 per 100,000 inhabitants in 2012-2016 (Goldbrunner et al., 2021). Among all cases, around 90% of meningiomas are classified as benign tumors (Fisher et al., 2021).
Several factors can be correlated with the impact of meningiomas, such as
- Advanced age,
- ionizing therapy,
- genetic susceptibility,
- Head trauma
- and the use of a contraceptive treatment (Lee and Lee, 2020).
Meningioma is more common in women (Wiemels et al., 2010). This could be correlated with higher progesterone receptors in women (Baldi et al., 2018).
Previous studies have already pointed out that progesterone receptors, estrogens and androgens are expressed in various types of meningiomas (Qi et al., 2013).
About 88 % of meningiomas had progesterone receptors, 40 % of estrogen receptors and 39 % were positive for androgens receptors (Korhonen et al., 2006). To date, several cases and cohorts have shown an increased risk of meningioma in women who use hormonal contraception (Harland et al., 2018).
Meningioma could occur in certain specific places influenced by their genetic profile (Van Den Munckhof et al., 2012). A previous study has reported that exogenous factors such as hormonal treatment also play a role in the development of meningioma in a specific place (APRA et al., 2020).
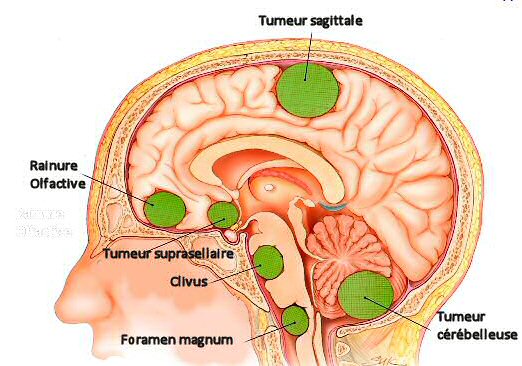
The study revealed that sphero-orbital meningioma has developed in women who have received progesterone treatment, and most of the cases were found in their fifties. Spheno-orbital locations include the large sphenoid wing with a distinct periorbital extension (Terrier et al., 2018). The most significant symptom of spheno-orbital meningioma is a visual disruption in 95% of cases and could occur with paresis of oculomotor nerves. Surgery is necessary in most cases with symptoms, but as their location can make complete resection difficult, adjuvant radiotherapy is recommended in certain cases (Terrier et al., 2018).
This study aimed to analyze the association between the history of the use of hormonal contraceptives and the location of meningiomas in the Indonesian population.
Materials and methods
This study collected retrospective data from medical records of all patients with confirmed meningiomas histologically at the Dr. Sardjito, Yogyakarta, Indonesia, 2019 to early 2021. As the history of hormonal contraception is the main variable of this study, only female patients have been included. In addition, patients without data on the use of hormonal contraceptives have been excluded. Due to the limited data of the medical file concerning the details of the use of hormonal contraception, data on the type of hormonal contraception and the duration of use of contraception was not available. Consequently, we only divided the patients in two groups: those with history of hormonal contraception at any time in their lives and those who do not have such history.
We have also collected data on age, symptoms, results of the pathology (WHO classification and histological type) and MRI or brain scanner before surgery (location and size of meningioma). The measurements of the dimension of the tumor were carried out during the last imaging examination before surgery. Images of Tomodensitometry with contrast and MRI weighted in T1 were used, but in the absence of contrast administration, the measures were still carried out provided that the examiner could identify the limit of the lesion with confidence. After the Recist measurement, the longest diameter of the tumor in the axial plane was recorded. The second diameter perpendicular in the axial plane was also taken to allow the compatibility of the measurement of the tumor according to the WHO. The third longest craniocaudal diameter was taken in the sagittal or coronal plane. The measure included any component of the tumors, that is to say the necrotic zone, the calcification, the cystic component and the intra-dusy part. The size of the tumor was determined by multiplying the two longest diameters on the imaging results.
All data were analyzed using IBM SPSS statistics version 26. A bivariate analysis was carried out to analyze the association between the use of hormonal contraceptives and the location of meningiomas. We used an independent T test or the Mann-Whitney test for digital data and the Test of the Chi square or the exact Fisher test for category data. A multivariate analysis using logistics regression with the retrograde method was then carried out to identify the variables independently associated with the location of meningiomas.
Results
The demographic and clinical characteristics of patients are presented in Table 1. Among the 99 patients included in this study, 72 (72.7 %) had history of hormonal contraception, while 27 (27.3 %) did not.
The average age at the time of the diagnosis of meningioma was 47.92 ± 8.29 years. There was no significant age difference between groups with and without hormonal contraception (p = 0.875).
The subjects consist of 83 (83.8%) OMS grade I and 16 (16.2%) tumors of Grade II and III of WHO. The most frequent histological types were meningothelial (33.3 %) and transitional (24.2 %). There was no statistically significant difference in the WHO classification and the histological type between hormonal contraception groups (p = 0.129 and 0.112, respectively). We did not find a statistical difference between hormonal contraception and the size of the tumor (p = 0.974).
We did not find a difference in the performance scale of Karnofsky (KPS) to the intake between the two groups (p = 0.517). Patients in the hormonal contraception group presented significantly more visual symptoms than those without history of hormonal contraception (66.7 vs 44.4% respectively, p = 0.044).
A bivariate analysis was carried out to analyze the association between various variables and the location of meningiomas (Table 2). A total of 57 (57.6 %) tumors were located in the spheno-organ, and 42 (42.4 %) were distributed in other locations. There was no significant difference between the two groups with regard to age at the time of diagnosis (p = 0.779), the histological type (p = 0.126) and the size of the tumor (p = 0.772).
As expected, patients with spheno-orbital meningioma had more visual symptoms than the group of tumors located in other locations (78.9 % against 35.7 %, p <0.001). In addition, patients with spheno-orbital meningioma also presented less crises (p <0.001) and fewer changes in speech (p = 0.039).
A significant association was found between the rank of meningiomas and their location, with a higher proportion of MENINGIMES of grade I of WHO in spheno-orbital tumors than in the meningiomas of other locations (91.2 vs 73.8%, p = 0.02).
We found a significant difference in the location of tumors between contraception groups (p = 0.038). Patients with history of hormonal contraception presented a higher proportion of meningiomas in sphero-orbital locations than patients without these history . (63.9 vs. 40.7% respectively, p = 0.038). A multivariate analysis was then carried out to confirm this result. The variables that could potentially affect the location of meningiomas and whose p <0.25 in the bivariate analysis were included. The result of the logistical regression test using the retrospective method is presented in Table 3. In this analysis, the only variable significantly associated with the location of meningiomas in the Spheno-Orbital Region was the use of hormonal contraceptives (ODDS Ratio (OR) 2.573, p = 0.041).
Table 1. Demographic and clinical characteristics of subjects (n = 99)
| Variable | Total | Hormonal contraception | P-Value | ||||
| No yes | |||||||
| Number of patients, n (%) | 99 (100) | 27 (27.3) 72 (72.7) | 72 (72.7) | ||||
| Average age, years (sd) | 47.92 (8.29) | 48.52 (9.18) | 46.38 (6.86) | 0.875* | |||
| WHO grade, n (%) | |||||||
| Grade i | 83 (83.8) | 20 (74.1) | 63 (87.5) | 0.129** | |||
| Others | 16(16.2) | 7 (25.9) | 9 (12.5) | ||||
| Histological type, n (%) | |||||||
| Meningothelial | 33 (33.3) | 9 (33.3) | 24 (33.3) | 0.112** | |||
| Transitional | 24 (24.2) | 9 (33.3) | 15 (20.8) | ||||
| Fibroblastic | 11 (11.1) | 0 (0) | 11 (15.3) | ||||
| Microcystic | 10 (10.1) | 1 (3.7) | 9 (12.5) | ||||
| Atypical | 13 (13.1) | 5 (18.5) | 8 (11.1) | ||||
| Other | 8 (8.1) | 3 (11.1) | 5 (6.9) | ||||
| Size in CM2, average (SD) | 28.68 (19.06) | 27.66 (16.27) | 29.07 (20.16) | 0.974* | |||
| Symptoms, n (%) | |||||||
| Headache | 82 (82.8) | 24 (88.8) | 58 (80.6) | 0.388** | |||
| View disturbances | 60 (60.6) | 12 (44.4) | 48 (66.7) | 0.044 | |||
| Epilepsy crisis | 21 (21.2) | 8 (29.6) | 13 (18.1) | 0.21 | |||
| Unconsciousness | 8 (8.1) | 2 (7.4) | 6 (8.3) | 1** | |||
| Personality change | 6 (6.1) | 2 (7.4) | 4 (5.6) | 0.663** | |||
| Modification of speech | 15 (15.2) | 6 (22.2) | 9 (12.5) | 0.344** | |||
| Cognitive changes | 6 (6.1) | 2 (7.4) | 4 (5.6) | 0.663** | |||
| Anomaly of the process | 2 (2) | 0 (0) | 2 (2.6) | 1** | |||
| Hemiparisia | 18 (18.2) | 8 (29.6) | 10 (13.9) | 0.084** | |||
| Nausea/vomiting | 6 (6.1) | 2 (7.4) | 4 (5.6) | 0.663** | |||
| KPS in admission, average (SD) | 72.61 (20.37) | 77.14 (17.36) | 69.81 (22.71) | 0.517* | |||
*, Mann-Whitney Test; **, Fisher exact test; ***, independent t-test, other tests use chi square test
Discussion
Previous studies have suggested that the development of meningiomas is particularly influenced by female sex hormones (Bernat et al., 2015). Our study has shown that around 70 % of women with meningioma had history of hormonal contraception. In line with ours, an anterior case study in Sweden found a high risk of meningioma with the use of hormonal contraception (Wigertz et al., 2006). The most used hormonal contraception in this study was injections containing high -dose progesterone.
These results comply with those of a clinical study conducted in Paris, in France, which revealed that spherorbital meningiomas are preferentially developing in women who follow hormone treatment towards the fifties (APRA et al., 2020).
One of the largest cohort studies in France also revealed that the risk of meningioma was much higher in women treated by cyproterone acetate (ACP), a product of progesterone synthesis, with a cumulative dose of more than 60 g. However, after a year of stopping treatment, the risk of meningioma has decreased significantly (Weill et al., 2021).
Another cohort study in French has shown a different distribution of ages among women who took ACP compared to the non-ACP population. Women who took ACP had a median age at the time of the 14-year-old meningioma surgery to that of the group without ACP (Champeaux-Deond et al., 2021) . At the same time, in our study, there was a slight average age difference between the population under hormonal contraception (46.38 ± 6.86 years) and that without hormonal contraception (48.52 ± 9.18 years). However, the difference was not statistically significant.
Previous studies have shown the possible association between the use of hormonal contraception and the location of meningiomas, in particular in the base of the skull (Peyre et al., 2018). APRA (2020) has found that sphero-orbital meningiomas are developing more often in women under hormonal contraception.
The study by Champeaux-Deond (2021) has shown that the meningiomas induced by hormonal contraception were preferentially located on the anterior and average skull base. Likewise, Peyre (2018) reported a predominance of the base of the previous skull in patients with meningiomas having taken hormonal contraception. In this study, we found a spheno-orbital meningiom in 57 (57.6%) case. Statistically, we have found a significant association between the use of hormonal contraceptives and the location of meningioma in the spheno-orbital region in bivariate analysis (p = 0.038). A multivariate analysis confirmed this association, showing that patients with history of hormonal contraception were twice and a half times more likely to develop meningioma in the spheno-orbital region as patients with no history of hormonal contraception (p = 0.041). However, the underlying mechanism is not clearly understood. This is probably linked to the higher level of progesterone receptors (PR) of meninges in this region. Previous studies have shown that meningiomas of the median skull base have a significantly higher number of cases with a strong expression of PR (APRA et al., 2020; Maiuri et al., 2021). However, there is a lack of evidence associating the quantity of hormonal receptor with tumor growth under treatment (APRA et al., 2020). This point requires a more in -depth study.
A greater visual impairment in patients with hormonal contraception of contraception could be linked to the location of the tumor in this spheno-orbital region resulting in compression of the optic nerve. Indeed, our study showed that out of 72 patients with history of hormonal contraception, 48 (66.7%) of them had a visual impairment (p = 0.04). Almost 80 % of patients with spheno-orbital meningioma presented a visual impairment, which is much higher than meningiomas of other locations (35.7 %) (p <0.001). A meta-analysis study had reported that the most present symptoms in sphening-organic meningiomas were proptosis (84%), unilateral visual impairment (46%) and the deficit in the visual field (31%) (Fisher et al., 2021). Consequently, the first -line treatment is surgery due to the compression of the optic nerves (Honeybul et al., 2001). In addition, spheno-orbital meningioma is also known to have a higher recurrence rate than meningiomas in other locations (Terrier et al., 2018).
The main limit of this study is the lack of information on duration, doses or the type of hormonal contraception used. Consequently, a more in -depth study to explore the association of these variables with the location of meningiomas in the Indonesian population is necessary. Another limit is the small number of meningiomas of Grade II and III of the WHO in our study.
In conclusion, the use of hormonal contraception is associated with meningiomas in the Spheno-Orbital region. The result is a higher number of patients with visual symptoms in the group with history of hormonal contraception.
Table 2. Bivariate analysis of the location of meningiomas
| Variable | Total | Location | P-Value | ||||
| Other spheroorbital | |||||||
| Number of patients, n (%) | 99 (100) | 57 (57.6) | 42 (42.4) | ||||
| Average age, years (sd) | 47.92 (8.3) | 47.89 (8.5) | 47.95 (8.04) | 0.779* | |||
| WHO grade, n (%) | |||||||
| Grade i | 83 (83.8) | 52 (91.2) | 31 (73.8) | 0.02 | |||
| Others | 16 (16.2) | 5 (8.8) | 11 (26.2) | ||||
| Histological type, n (%) | |||||||
| Meningothelial | 33 (33.3) | 22 (38.6) | 11 (26.2) | 0.126** | |||
| Transitional | 24 (24.2) | 14 (24.6) | 10 (23.8) | ||||
| Fibroblastic | 11 (11.1) | 8 (14) | 3 (7.1) | ||||
| Microcystic | 10 (10.1) | 6 (10.5) | 4 (9.5) | ||||
| Atypical | 13 (13.1) | 3 (5.3) | 10 (23.8) | ||||
| Other | 8 (8.1) | 4 (7.1) | 4 (9.5) | ||||
| Size in CM2, average (SD) | 28.68 (19.06) | 28.29 (20.04) | 29.17 (18.03) | 0.772* | |||
| Symptoms, n (%) | |||||||
| Headache | 82 (82.8) | 45 (78.9) | 37 (88.1) | 0.233 | |||
| Visual disturbances | 60 (60.6) | 45 (78.9) | 15 (35.7) | <0.001 | |||
| Epilepsy crisis | 21 (21.2) | 4 (7.1) | 17 (40.5) | <0.001 | |||
| Unconsciousness | 8 (8.1) | 3 (5.3) | 5 (11.9) | 0.278** | |||
| Personality change | 6 (6.1) | 2 (3.5) | 4 (9.5) | 0.397** | |||
| Change of speech | 15 (15.2) | 5 (8.8) | 10 (23.8) | 0.039 | |||
| Cognitive change | 6 (6.1) | 1 (1.8) | 5 (11.9) | 0.08** | |||
| Anomaly of the process | 2 (2) | 1 (1.8) | 1 (2.4) | 1** | |||
| Hemiparisia | 18 (18.2) | 7 (12.3) | 11 (26.2) | 0.076 | |||
| Nausea/vomiting | 6 (6.1) | 2 (3.5) | 4 (9.5) | 0.397** | |||
| KPS in admission, average (SD) | 72.61 (20.37) | 72.45 (21.2) | 72.82 (19.46) | 0.819* | |||
| Use of hormonal contraceptives, n (%) | |||||||
| Yes | 72 (100) | 46 (63.9) | 26 (36.1) | 0.038 | |||
| No | 27 (100) | 11 (40.7) | 16 (59.3) | ||||
*, Mann-Whitney Test; **, Fisher exact test; ***, independent t-test, other tests use chi square
Table 3. Multivariate analysis by logistical regression of factors affecting the location of meningiomas
| Variable | Odds Ratio | 95 % CI | P-Value |
| Use of hormonal contraceptives | 2.573 | 1.040-6.367 | 0.041 |
Author's declaration of contribution
RGM and EKD formulated the idea presented and designed the study. RAH, DNFF, KRD, AR, MAS, ASW, KD, EBS, AA and RGM collected samples and clinical data. NHS analyzed all MRI and CT exams. RGM and EKD have developed the theory, carried out the statistical analysis and wrote the initial version of the manuscript. All the authors discussed the results and contributed to the final manuscript. RGM has prepared the final manuscript. All projects were supervised by EKD.
Thanks
This research was supported by the PDUPT grant from the Indonesian Ministry of Research and Higher Education, number 8/E1/KPT/2021 and 3572/E4/AK.04/2021 in EKD.
Ethical approval
This study obtained the ethical approval of the Institutional Review Board (IRB), Faculty of Medicine, Public Health and Nurses, Universitas Gadjah Mada, Indonesia. The written informed consent was obtained from the patients themselves or a member of their family.
Conflicts of interest
The authors have no conflict of interest to declare.
References
APRA C, Roblot P, Alkhayri A, et al (2020). Female gender and exogenous progesterone exhibition as risk factors for spheno-organ meningiomas. J Neurooncol, 149, 95-101.
Baldi I, Engelhardt J, Bonnet C, et al (2018). Epidemiology of meningiomas. Neurosurgery, 64, 5-14.
Bernat Al, Oyama K, Hamdi S, et al (2015). Growth Stabilization and Regression of Meningiomas after Discontinuization of Cyproterone Acetate: A Case Series of 12 Patients. Acta Neurochir, 157, 1741-6.
Champeaux-Deland C, Weller J, Froelich S, Sartor A (2021). Cyproterone acetate and meningioma: a nationwide-wide population based study. J Neurooncol, 151, 331–8.
Fisher FL, Zamanipoor Najafabadi Ah, Schoones JW, Genders SW, Furth WR (2021). Surgery as a safe and effective Treatment option for spheno-orbital meningioma: A Systematic Review and Meta-Analysis of Surgical Techniques and Outcomes. Acta Ophthalmol, 99, 26-36.
Fisher Fl, Zamanipoor Najafabadi Ah, van der Meer Pb, et al (2021). Long-Term Health-Related Quality of Life and Neurocognitive Functioning After Treatment in Skull Base Meningioma patients. J neurosurg, 1, 1-13.
Goldbrunner R, Stavrinou P, Jenkinson MD, et al (2021). Eano Guideline on the Diagnosis and Management of Meningiomas. Neurooncol, 23, 1821-34.
Harland TA, Freeman JL, Davern M, et al (2018). Progesteroneonly contraception is associated with a shorter progressionfree survival in premenopausal women with who grade i meningioma. J Neurooncol, 136, 327-33.
Honeybul S, Neil-Dwyer G, Lang Da, Evans BT, Ellison DW (2001). Sphenoid Wing Meningioma in plate: A Clinical Review. Acta Neurochir, 143, 749-58.
Korhonen K, Salminen T, Raitanen J, et al (2006). Female predominance in meningiomas can not be explained by different in progesterone, estrogen, or Androgen receptor expression. J Neurooncol, 80, 1-7.
Lee YS, Lee YS (2020). Molecular characteristics of meningiomas. J pathol transl med, 54, 45.
Mauri F, Mariniello G, from Divitiis O, et al (2021). Progesterone Receptor Expression in Meningiomas: Pathological and Prognostic Implications. Front Oncol, 11, 2585.
Ostrom QT, Gittleman H, Truitt G, et al (2018). CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2011–2015. Neurooncol, 20, IV1-86.
Peyre M, Gaillard S, de Marcellus C, et al (2018). Progestinassocated shift of Meningioma Mutual Landscape. Ann Oncol, 29, 681-6.
Qi Zy, Shao C, Huang Yl, et al (2013). Reproductive and exogenous hormone factors in Relation to risk of meningioma in women: a meta-analysis. PLOS One, 8, E83261.
Terrier LM, Bernard F, Fournier HD, et al (2018). SPHENO-ORBITAL MENINGIOMAS SURGERY: MULTICENTER Management Study for Complex Extensive Tumors. World Neur, 112, E145-56
Van den Munckhof P, Christiaans I, Kenter SB, Baas F, Hulsebos TJM (2012). Germline SMARCB1 PREDISPOSES TO MULTIPLE MENINGIOMAS AND SCHWANNOMAS with preferential rental of Cranial meningiomas at the FALX CEREBRI. Neurogenetics, 13, 1-7.
Weill A, Nguyen P, Labidi M, et al (2021). Use of High dose Cyproterone Acetate and Risk of Intracranial Meningioma in Women: Cohort Study. BMJ, 372.
Wiemels J, Wrensch M, Claus EB (2010). Epidemiology and Etiology of Meningioma. J Neurooncol, 99, 307-14.
Wigertz A, Lönn S, Mathiesen T, et al (2006). Risk of Brain Tumors Associated With Expores To Exogess Female Sex Hormones. Am J Epidemiol, 164, 629-36.